Effect of Pre-Treatment and Drying Methods on the Antioxidant Properties and Amino Acid Profile of Tomato Powder
- 1. Centre for food Technology and Research, Benue State University, Nigeria
- 2. Departments of Agriculture, Food Technology Services, Nigeria
- 3. Department of Food Science and Technology, University of Agriculture, Nigeria
- 4. Akperan Orshii College of Agriculture, Benue State University, Nigeria
Abstract
Processed fruits and vegetables have been long considered to have lower nutritional value than their fresh commodities due to the loss of vitamins during processing. Thirty-six kilograms of freshly harvested tomatoes (UTC variety) were purchased from a Kaduna Central Market. Three pre-treatments were used namely: 0.2g of potassium metabisulphite (K2 S2 O) per 100 g of tomato, blanching at 60°C for 1 min and control (untreated) samples. Freeze, hybrid-solar and oven drying were employed and processed in powder. Antioxidant activities and free amino acids have been evaluated. All the tomato powders showed better antioxidant activities than the synthetic standards. In terms of the pre-treatments employed, the antioxidant activities of the powders were observed to be in the order of potassium metabisulphite>blanched>control. As for drying methods the order was as observed as follows freeze>oven>solar-hybrid dried. branched chain amino acids (BCAAs) content in tomato powders ranged from 1.327 to 1.583% with the solar-hybrid dried sample pre-treated with potassium metabisulphite has the highest (1.583%) compared to the lowest value (1.316%) in the freeze dried sample pre-treated with potassium metabisulphite. Like was with the content of BCAA, Hydrophobic amino acids (HAA) content was highest (3.905%) in the K2 S2 O treated hybrid-solar dried tomato powder sample whereas the lowest value (3.393%) was recorded by the K2 S2 O treated oven dried sample. The Negatively charged amino acid (NCAA) were the most abundant amino acids in the tomato powder samples ranging in content from 5.272 to 7.949% with the control freeze dried sample having the highest content (7.949%) and the K2 S2 O treated oven dried sample the least (5.272%). Therefore it was concluded that the antioxidant and amino acid characteristics of UTC tomato powder is significantly affected by different pretreatment and drying methods. This information may have a significant impact on consumers’ food selection by increasing their consumption of tomato powder to reduce the risks of chronic diseases.
Keywords
• Post-harvest losses
• Tomato
• Powder
• Antioxidant properties
• Amino acid
Citation
Drammeh L, Girgih AT, Ahemen SA (2018) Effect of Pre-Treatment and Drying Methods on the Antioxidant Properties and Amino Acid Profile of Tomato Powder. J Hum Nutr Food Sci 6(2): 1126.
ABBREVIATIONS
DPPH: Diphenyl?1?Picrylhydrazyl; FRAP: Ferric Reducing Antioxidant Power; FICA: Ferrous Ion Chelating Ability; SRSA: Superoxide Radical Scavenging Activity
INTRODUCTION
Fresh tomato is perishable and to extend its shelf life one form of processing or the other is needed. This reduces postharvest losses and ensures the availability of tomato year round [1]. Drying is one of the oldest methods of food preservation used by human beings and also the most widely used method for food preservation purposes. During drying processes, water in food products is reduced to a level where the growth of spoilage microorganisms, as well as the occurrence of chemical reactions, are halted or slowed down. In addition to preservation, the reduced weight and bulk volume of dried products and their longer shelf stability reduce the costs and/or difficulties of product packaging, handling, storage, and distribution [24]. Heating drying processes may cause undesirable changes in tomato composition and loss of quality, mainly in bioactive components [5]. Pre-treatments are common in most of the drying processes in order to improve product quality, storage stability, and process efficiency. In recent years, an improvement in quality retention of the dried products by altering processing strategy and pre-treatment has gained much attention. A variety of non-enzymatic chemical changes taking place in certain materials during drying also affect adversely the quality of the finished product [6].
Therefore; in view of the above benefits of pre-treating tomato with blanching and potassium metabisulphite; conversion of tomato into a value added product like tomato powder will be very useful. The objective of this research therefore was to produce tomato powder and evaluate the effect of pre-treatment and drying methods on antioxidant and amino acid qualities of tomato with the view of maintaining antioxidants and amino acids which play an important role in the management of generative diseases as well as controlling the post- harvest losses of this valuable plant fruit.
MATERIALS AND METHODS
Materials
The tomato (UTC variety) for this research work was bought at Kaduna Central Market, Kaduna State, Nigeria.
Methods
Pre-treatment method:
Control: 4 kg tomato slices was directly dried without any pre-treatment prior to drying
Potassium metabisulphite: 4 kg of tomato slices was completely soaked in a solution of potassium metabisulphite (K2S2O) 0.2g/100g for 10 min prior to drying.
Blanching: 4 kg tomato slices was dipped in a hot water (70°C) for 1 min and drained using a muslin cloth for 5 min prior to drying.
Drying method
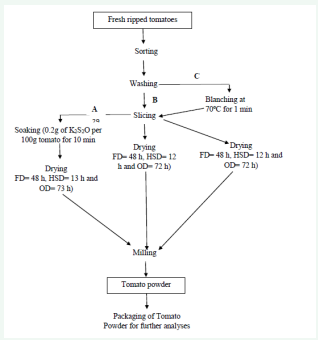
Freeze drying: 4 kg tomato slices per pre-treatment was dried in a freeze drier (Sikuan LGJ-10, Beijing, China) at Kaduna Polytechnic (0.25 kg on each tray) at -30°C which falls within range of - 45 and -20°C as recommended by Sablani and Rahman [7]. The drying time required to reach the equilibrium moisture content were 2880 min (48 h) for all the samples and the moisture content of the dried slices was 6.21, 6.22 and 6.23 % for powders pre-treated with KMS treated, blanched and untreated samples, respectively (Figure 1).
Figure 1: Production of tomato powder
Source: Mozumder et al. [16], with slight modifications 43
A: Potassium metabisulphite treated; B: Control; C: Blanched 44
Solar-hybrid drying: 4 kg of tomato slices per pre-treatment was dried in the constructed solar-hybrid dryer using two heating source (solar and kerosene stove) together at Nigerian Stored Product Research Institute (NSPRI). The temperature of the hybrid drier was 53°C prior to loading the tomato slices. 1 kg of sliced tomatoes per tray was then loaded in the drier. The slices were weighed after first 8 h and weighed after every 1 h until the weight remained constant after three consecutive reading. The drying time required to reach the equilibrium moisture content were 720, 720 and 780 min and the moisture content of the dried slices was 8.86, 8.88 and 8.90 % for powders pre-treated with KMS treated, blanched and untreated samples, respectively. The average of atmospheric temperature was approximately 35 to 43°C daily.
Oven drying: 4 kg of tomato slices per pre-treatment was dried in an oven (Genlab T12H, UK) at Kaduna Polytechnic (0.5 kg on each tray) at 60°C which falls within range of 60-66°C as recommended by DeLong [8]. The oven was pre-heated to 60°C prior to loading the UTC tomato slices. The slices were weighed after first 78 h and weighed after every 6 h until the weight remained constant after three consecutive reading. The drying time required to reach the equilibrium moisture content were 4320 min (72 h), 4320 min (72 h) and 4380 min (73h) and the moisture content of the dried slices was 7.17, 7.20 and 7.24 % for powders pre-treated with potassium metabisulpite treated, blanched and untreated samples, respectively (Figure 2).
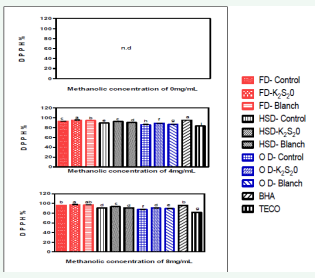
Figure 2: DPPH radical scavenging activity of tomato powder at different methanolic concentration
Abbreviations: Prefix FD: Freeze Dried; Prefix HSD: Hybrid-Solar Dried; Prefix OD: Oven Dried; K2S2O: Potassium Metabisulphite; BHA: Butylated Hydroxyanisole; TECO= α-tocopherol; n.d: Not Detected; DPPH: α,α-Diphenyl-1-picrylhydrazyl
Antioxidant properties
Determination of 2,2-Diphenyl-1-picrylhydrazyl (DPPH) radical scavenging activity, Ferric reducing antioxidant power, Ferrous ion chelating ability and Superoxide radical scavenging activity were carried out as done by Chang et al. [10].
DPPH radical scavenging activity (DRSA)
Different weighted tomato powder samples (100–1000 mg) were mixed with 50 mL methanol to prepare samples with the weight to volume ratios of 0, 4, and 8, mg/mL, respectively. 5 mL of each test solution, including MEs of tomato samples, α-tocopherol, and BHA solutions, was mixed with 1 mL of 1 mM freshly prepared DPPH methanolic solution, and left to stand for 30 min prior to being spectrophotometrically detected at 517 nm. The percentage of DPPH scavenging activity is expressed by:
DPPH radical scavenging activity
Where: Ab = absorbance of the blank
As = absorbance of the sample
Ferric reducing antioxidant power (FRAP)
Different weighted tomato powder samples (100-1000 mg) were mixed with 50 mL methanol to prepare samples with the weight to volume ratios of 0, 4, and 8, mg/mL, respectively. 10 mL of the above samples along with methanolic solutions of α-tocopherol and BHA were filtered in vacuum and then mixed with 2.5 mL phosphate buffer (0.2 M, pH6.6) and 2.5 mL of 1% potassium ferricyanide. The mixture was incubated in a 50°C water-bath for 20 min, then rapidly cooled, mixed with 2.5 mL of 10% trichloroacetic acid for 5min, and centrifuged at 3000g for 10 min. 5 mL of the supernatant mixed with 5 mL DI water and 1 mL of 0.1% ferric chloride, and left to stand for 10 min. Absorbance at 700 nm was used as the indicator of reducing power.
Ferrous ion chelating ability (FICA)
Different weighted tomato powder samples (100–1000 mg) were mixed with 50 mL methanol to prepare samples with the weight to volume ratios of 0, 4, and 8, mg/mL, respectively. 5 mL of the test solutions, including MEs of tomato samples, α-tocopherol, and BHA, solutions were mixed with 0.1 mL of 2 mM FeCl2 and 0.2 mL of 5 mM ferrozine solutions and allowed to react for 10 min. The absorbance at 562 nm of the resulting solutions were measured and recorded. The percentage of ferrous ion chelating ability is expressed by:
Where: Ab = absorbance of the blank
As = absorbance of the sample
Superoxide radical scavenging activity (SRSA)
The effect of the methanolic extract on superoxide anion radicals was estimated according to the method described by Talaz et al. [11], with slight modifications. The reaction mixture containing 1 mL of riboflavin (3.3 × 10 mol/L), methionine (0.01 mol/L), 1 mL of nitro blue tetrazolium chloride (NBT, 4.6 × 10 mol/L), and the solvent of the above solutions was phosphate buffer solution (0.05 mol/L, pH 7.8). After adding 1 mL of samples and ascorbic acid as a control of different concentrations of methanolic extracts (0, 4 and 8, mg/mL), the reaction mixture was illuminated at 4000 lx and 25°C for 30 min. Then the absorbance of the reaction mixture was measured at 560 nm with a spectrophotometer (Spectro UVS-2700, Labomed, and Culver City, CA, USA) and the scavenging percentage was calculated according to the following formula:
Where Ab = the absorbance values of the blank
As= the absorbance values of the sample
Amino acid composition analysis
Tomato powder samples were hydrolysed for 24 h with 6 M HCl followed by analytical HPLC detection and quantification of amino acids according to the method of Bidlingmeyer et al. [12]. To determine cysteine and methionine contents, performic acid oxidation was used instead of HCl [13], while tryptophan was determined after alkaline hydrolysis [14]. Statistical analysis
The data were presented as the mean of three determinations ± standard deviation. Data were analyzed by one-way analysis of variance (ANOVA) and Duncan’s multiple-range test by using Statistical Package for the Social Sciences Statistical Analysis (SPSS) Version 20. P values ≤ 0.05 were regarded as significant [15,16].
RESULTS AND DISCUSSION
Diphenbyl-1-picrylhydrazyl
Antioxidant activity of natural compounds has been shown to be involved in termination of free radical reactions [17]. Diphenbyl-1-picrylhydrazyl (DPPH) is a stable free radical and accepts an electron or hydrogen radical to become a stable diamagnetic molecule. The model of scavenging DPPH radical is especially used to evaluate chain-breaking activity in the propagation phase of lipid (and protein) oxidation [18]. The effect of antioxidants on, DPPH radical scavenging was thought to be due to their hydrogen donating ability [19]. When antioxidant reacts with this stable radical, the electron becomes paired off and bleaching of the colour stoichiometrically depends on the number of electrons taken up [20]. As shown on figure 3, the methanolic extract (ME) from various processed tomato powders, along with α-tocopherol and BHA all showed excellent scavenging effect on DPPH radicals; they were in the range of 82.33–97.33% at concentration higher than 4 mg/mL.
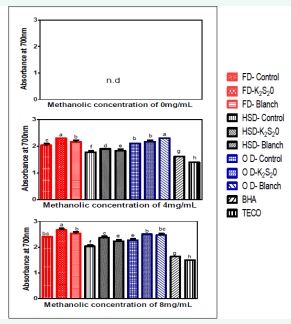
Figure 3: Ferric reducing antioxidant power of tomato powder at different methanolic concentrations
Abbreviations: Prefix FD: Freeze Dried; Prefix HSD: Hybrid-solar Dried; Prefix OD: Oven Dried; K2S2O: Potassium Metabisulphite; BHA: Butylated Hydroxyanisole; TECO= α-tocopherol; n.d: Not Detected.
The tomato powders treated with K2 S2 O gave the highest DPPH radical scavenging activity followed by blanching and control tomato powder in all the drying methods. The extract from OD-UN showed the lowest scavenging activity of 85%. This activity increased with an increase in the concentrations of the samples from 4 to 8 mg/mL, only the K2 S2 O treated freeze dried sample at 8 mg/ mL of methanolic concentration reached a scavenging activity comparable to BHA. Meanwhile, α-tocopherol exhibited the lowest DPPH radical scavenging activities amongst the samples. In comparison with the results of Yang et al. [21], DPPH radical scavenging effects of 2 mg/mL MEs from commercial mushrooms were 42.9–69.9%. Compared to that, tomato powder is considered to have much higher DPPH radical scavenging activity than that from commercial mushrooms Wong and Yen [22], the DPPH radical scavenging activity at 2 mg/mL MEs of tomato was higher than that at the same concentration of mungbean and soybean sprouts, and equal to that of radish sprouts. El-Hamidi and Shami [23] reported that the DPPH activity of garlic powder extracts at MEs concentration of 15 mg/mL was at a range of 16.39-19.47% which when compared to tomato powders at MEs concentration of 8 mg/mL was at a range of 87-98%. This clearly indicated that tomato powder has a better DPPH activity than garlic powder.
Ferric reducing antioxidant power
The ferric reducing antioxidant power (FRAP) mechanism is based on electron transfer rather than hydrogen atom transfer [24]. In the FRAP assay, a ferrous-tripyridyltriazine (Fe3+- TPTZ) complex is reduced to its ferrous form, which absorbs light of 593nm. The change from the colourless ferric (Fe3+) form to the intensely blue-coloured ferrous (Fe2+) form is caused by the combined action of electron-donating antioxidant in the test sample [25]. However, potential problems for its application in foods exist; as many metal chelators in food extract could bind Fe3+ and form complexes that also react with antioxidants [26]. The ferric reducing antioxidant powers of MEs of tomato powders along with α-tocopherol and BHA are shown in Figure (3). The FRAP of all the tomato powder samples were raised with the increase of MEs concentration. At concentration up to 4 mg/ mL, the reducing powers in all tomato samples were higher than that of α-tocopherol and BHA. All the tomato powders exhibited excellent FRAP values. At the same concentration, the control hybrid-solar dried sample (1.77%) showed the lowest reducing power. The K2 S2 O treated freeze dried tomato powder (2.67%) showed the highest while the ME concentrations were at 8 mg/ mL. It was observed that the tomato powders pre-treated with K2 S2 O treated and blanching exhibited higher FRAP than the control samples. Pinela et al. [27], worked on the FRAP of freeze dried tomato powder from four cultivars which had a range of 1.63-2.04%. When compared with this study, the all freeze dried tomato powders were observed to have FRAP (2.3-2.7%), which indicates to have a higher value than the latter; which could be due to the cultivar difference of the tomatoes from which the powders were obtained. Siddhuraju et al. [28], reported that the reducing power could be attributed mainly to the bioactive compounds associated with antioxidant activity. These bioactive compounds present in tomato’ MEs, including ascorbic acid, total phenols, lycopene, and other hydrophilic or hydrophobic antioxidants, are good electron donors and could terminate the free radical chain reactions by converting free radicals to more stable products. This antioxidant ability helps in the positive management of oxidative stress related ailments such as cancer, cardiovascular diseases etc. K2 S2 O treated showed the best FRAP property followed by the blanched and then the control samples.
Ferrous ion chelating ability
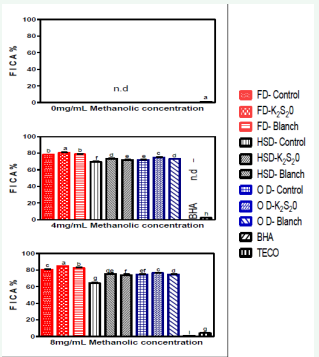
Metal chelating capacity is claimed as one of the antioxidant activity mechanism [29], since it reduces the concentration of the catalyzing transition metal in lipid peroxidation [30]. It is reported that chelating agents, who form α-bonds with metal, are effective as secondary antioxidants because they reduce the redox potential, thereby stabilizing the oxidized form of the metal ion [31]. As shown in Figure 4 Ferrous Ion Chelating Abilities (FICAs) of MEs from tomato powders were increased with the increase in MEs concentration while in lower concentration.
Figure 4: Ferrous ion chelating ability of tomato powder at different methanolic concentrations
Abbreviations: Prefix FD: Freeze Dried; Prefix HSD: Hybrid-Solar Dried; Prefix OD: Oven Dried; K2S2O: Potassium Metabisulphite; BHA: Butylated Hydroxyanisole; TECO= α-tocopherol; n.d: Not Detected.
The FICA values of all the tomato samples were kept in the range of 74.67–85.00% when the MEs concentration reached 8 mg/mL. However, the FICAs of methanolic solution of 8 mg/mL of BHA and α-tocopherol were 1.00% and 4.00%, respectively. This indicates that BHA and α-tocopherol have very little FICA. Since ferrous ions are the most effective prooxidants in the food system, the higher chelating abilities of MEs from tomato powders would be beneficial. As it can be seen, both BHA and α-tocopherol could hardly carry the FICA due to their chemical structure properties. MEs of Freeze, hybrid-solar and oven drying showed the highest FICA of 85%, 75% and 81% at 8 mg/mL, respectively. The FICA was highest with samples pretreated with K2 S2 O followed by blanching and control samples for all the drying methods. In literature, the MEs of various processed tomatoes (freeze-dried and hot-air-dried tomatoes) at a concentration of 2mg/mL could reach more than 65% FICA [32]. BHA and α-tocopherol had very little FICA. MEs at 8 mg/ mL of BHA and α-tocopherol were only 1% and 4%, respectively. Since ferrous ions are the most effective pro-oxidants in the food system, higher chelating abilities from tomato powder would be beneficial. It was reported by Srivastava and Kulshreshtha [22] that the use of a soybean sprout extracts at a concentration of 2 mg/mL was required to obtain 82% of FICA. Meanwhile, the MEs of mungbean sprouts and radish sprouts only exhibited a FICA of 58% and 36%, respectively, at a concentration of 2 mg/mL, when compared with this study, tomato powder was considered to contain a better FICA than that of mungbean sprouts and radish sprouts but soybean sprout exhibited better FICA results than tomato powder. In a study carried out by Yen et al. [33], it was reported that dried carrots at a MEs concentration of 2 mg/ mL all had FICA of above 80%, which had a higher values when compared to this study. This shows that carrot has a better FICA property than tomato powder (Table 1).
|
Table 1: Experimental design. |
|||
|
Pre-treatment
Drying method |
No pre-treatment (Control) |
K2S2O5 (0.2g/100g for 10 minutes) |
Hot Water Blanching (70°C for 1 Minute) |
|
Freeze drying (FD) [-30°C] |
Control |
FD- K2S2O |
FD-Blanched |
|
Hybrid-solar drying (HSD |
Control |
HSD- K2S2O |
HSD-Blanched |
|
Oven drying (OD) [60°C] |
Control |
OD- K2S2O |
OD-Blanched |
|
Experiment design as described by Mozumder et al. [16], with slight modification. |
|||
Key
Superoxide radical scavenging activity
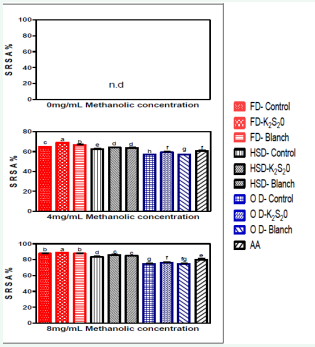
Superoxide is a highly reactive molecule that can react with many substrates produced in various metabolic processes including phagocytosis [34]. It can cause the oxidation or reduction of solutes depending on their reduction potential. Both aerobic and anaerobic organisms possess superoxide dismutase enzyme that can convert free-radical oxygen to molecular oxygen and peroxide or otherwise catalyses the breakdown of superoxide radical [35]. It can also reduce certain iron complex such as cytochrome [36]. Figure 5 shows the superoxide radical scavenging abilities (SRSAs) of MEs from tomato powders were increased with the increase in MEs concentration while in lower concentration.
Figure 5: Superoxide radical scavenging activity (SRSA) of tomato powder at different methanolic concentrations
Abbreviations: Prefix FD: Freeze Dried; Prefix HSD: Hybrid-Solar Dried; Prefix OD: Oven Dried; K2S2O: Potassium Metabisulphite; AA: Ascorbic Acid.
The SRSA values of all the tomato samples were kept in the range of 74.67–85.67% when the MEs concentration reached 8 mg/mL where the control oven dried sample recorded 74.64% (lowest) and the K2 S2 O treated freeze dried sample recorded 85.67% (highest). However, the SRSAs of methanolic solution of 8 mg/mL ascorbic acid as only 80.00%, which was only better than the entire oven, dried tomato powders. Ascorbic acid had more antioxidant activity than the control oven dried sample at all the levels of ME concentrations. The rest of the samples recorded better SRSA than AA. In a study on antioxidant properties of methanolic extract from Inula graveolens L. carried out by Al-Fartosy [37] reported that the scavenging activity was 82.51% at a concentration of 4 mg/ mL and 93.43% at a concentration of 12 mg/mL when compared to this study, Inula graveolens L. proves to have better SRSAs than tomato powder. A similar study done by Venkatachalam et al. [38], on Desmodium gangeticum when compared to this study, the tomato powders were found to have better SRSA than the Desmodium gangeticum.
Amino acid profile of tomato powder samples
Table 2 shows the amino acid (AA) composition of tomato powders pre-treated and dried using different methods.
|
Table 2: Amino acid composition of tomato powder samples. |
|||||
|
Amino |
FD- |
FD- |
FD- |
HSD- |
OD- |
|
Acid |
Control |
K2S2O |
Blanched |
K2S2O |
K2S2O |
|
ASX |
2.315 |
2.343 |
2.205 |
1.813 |
1.676 |
|
THR |
0.435 |
0.412 |
0.399 |
0.427 |
0.383 |
|
SER |
0.526 |
0.497 |
0.488 |
0.543 |
0.471 |
|
GLX |
5.634 |
5.372 |
5.351 |
4.024 |
3.596 |
|
PRO |
0.385 |
0.350 |
0.384 |
0.430 |
0.371 |
|
GLY |
0.482 |
0.446 |
0.454 |
0.512 |
0.447 |
|
ALA |
0.454 |
0.459 |
0.446 |
0.518 |
0.472 |
|
CYS |
0.224 |
0.203 |
0.211 |
0.206 |
0.172 |
|
VAL |
0.436 |
0.382 |
0.390 |
0.476 |
0.414 |
|
MET |
0.189 |
0.174 |
0.173 |
0.200 |
0.155 |
|
ILE |
0.376 |
0.354 |
0.348 |
0.436 |
0.371 |
|
LEU |
0.637 |
0.580 |
0.589 |
0.671 |
0.606 |
|
TYR |
0.361 |
0.312 |
0.323 |
0.344 |
0.294 |
|
PHE |
0.490 |
0.489 |
0.456 |
0.509 |
0.446 |
|
HIS |
0.796 |
0.667 |
0.816 |
0.587 |
0.613 |
|
LYS |
0.581 |
0.549 |
0.546 |
0.503 |
0.482 |
|
ARG |
0.608 |
0.535 |
0.543 |
0.609 |
0.487 |
|
TRP |
0.124 |
0.128 |
0.106 |
0.115 |
0.092 |
|
AAA |
0.975 |
0.929 |
0.885 |
0.968 |
0.832 |
|
BCAA |
1.449 |
1.316 |
1.327 |
1.583 |
1.391 |
|
HAA |
3.676 |
3.431 |
3.426 |
3.905 |
3.393 |
|
PCAA |
1.985 |
1.751 |
1.905 |
1.699 |
1.582 |
|
NCAA |
7.949 |
7.715 |
7.556 |
5.837 |
5.272 |
|
SCAA |
0.413 |
0.377 |
0.384 |
0.406 |
0.327 |
|
EAA |
4.064 |
3.735 |
3.823 |
3.924 |
3.562 |
|
Abbreviations: ASX: Aspartic Acid + Asparagine; GLX: Glutamic Acid + Glutamine; Combined Total of Hydrophobic Amino Acids (HAA): Alanine, Valine, Isoleucine, Leucine, Tyrosine, Phenylalanine, Tryptophan, Proline, Methionine and Cysteine; AAA: Aromatic Amino Acids: Phenylalanine, Tryptophan and Tyrosine; PCAA: Positively Charged Amino Acids: Arginine, Histidine, Lysine; NCAA: Negatively Charged Amino Acids: Aspartic + Asparagine, Glutamic + Glutamine, Threonine, Serine; SCAA: Sulphur Containing Amino Acids: Methionine, Cysteine; EAA: Essential Amino Acids; Sulphur Containing Amino Acid: Methione and Cysteine, Histidine, Isoleucine, Leucine, Lysine, Methionine, Phenylalanine, Threonine, Tryptophan and Valine |
|||||
The results of the preliminary screening for antioxidant properties of the tomato powder samples guided us towards the selection of five samples for evaluation of the amino acid content based their potency. As is evident from the AA composition results, the tomato powders contained all the twenty amino acids essential for synthesis of tissues and modulation of physiological activities. The amino acid composition of proteins and their derived peptides have been reported to possess a great influence on their functionality and bioactivity in vivo when consumed [39].
Aromatic amino acid (AAA) has been reported to possess strong radical scavenging potentials which protects against lipid peroxidation. The presence of tryptophan and tyrosine amino acids is associated with prevention of blindness resulting from the formation of cataract and find use in topical applications [40]. The control freeze dried sample had highest (0.975%) content of AAA while the K2 S2 O treated oven dried sample possessed the least (0.832%) content overall, all the evaluated samples have some degree of effect on reducing oxidant destruction cellular organelles. Branched chain amino acids (BCAAs) which include leu, Ile and Val are popular in the fitness industry where they are used during exercise to reduce fatigue, muscle soreness accelerate recovery and improve the use of fat for energy and can help reduce many chronic human diseases such as cancer, hypertension, liver, kidney diseases etc [41,42]. BCAA content in tomato powders ranged from 1.327 to 1.583% with the K2 S2 O treated hybrid-solar dried sample having the highest (1.583%) compared to the lowest value (1.316%) in the K2 S2 O treated freeze dried sample. Supplementation of BCAA in our diet is critical as it has several benefits towards the prevention and treatment of several morbidities in human beings. Hydrophobic amino acids (HAA) have been reported to be associated with good antioxidants properties, because of their ability to enhance interactions of peptides with lipids membrane bilayera in order to facilitate their entry into target organs and areas of need. This has been possible because of the structural nature of HAA which confers on them properties to achieve maximal interactions with the lipid moiety of lipid foods [43]. Like was with the content of BCAA, HAA content was highest (3.905%) in the K2 S2 O treated hybrid-solar dried tomato powder sample whereas the lowest value (3.393%) was recorded by the K2 S2 O treated oven dried sample. Positively charged amino acid (PCAA) including Arg, His and Lys are believe to accept electrons from negatively charged radical species thereby quenching their instability leading to the disruption of normal physiological functions of body organs. The imidazole ring found in the His structure is known to promote radical scavenging activities thereby reducing the destructive tendencies of free radicals and reactive oxygen species (ROS) and additionally, imidazole is known to an important proton donor [44]. The amount of PCAA in the tomato powder ranged from 1.751 to 1.985% with the control freeze dried sample have the highest content of 1.985% and the K2 S2 O treated freeze dried sample having the lowest content of PCAA. Negatively charged amino acid (NCAA) such as aspartic acid, asparagine, glutamic acid (ASX and GLX) has been shown to be donors of excess electrons to free radicals during redox reactions that help quench their harmful hyper-reactions [45]. The NCAA were the most abundant AAs in the tomato powder samples ranging in content from 5.272 to 7.949% with the control freeze dried sample having the highest content (7.949%) and the K2 S2 O treated oven dried sample the least (5.272%). The four commonly known sulfur-containing amino acids are methionine, cysteine, homocysteine and taurine but principally among them, methionine and cysteine are among he twenty traditional amino acids that are incorporated into proteins and sulfur-containing amino acids are more abundant in animal and cereal proteins than in legume proteins [46]. Cystine (a combination of two is a sulphur-containing amino acid that is required for the formation of healthy skin, hair, bones, and connective tissues as well as in make glutathione, one of the natural antioxidants that deactivates free-radical and prevent their damaging effects. Glutathione is also known to detoxify the liver. Sulphur-containing amino acids with good antioxidant abilities might be useful for the food processing industry as antioxidant additives to delay spoilage and prolong shelf-life of food products and may offer health beneficial pharmacological effect against oxidative stress cell damage [47]. Though the amount of sulphur containing AA was generally low in the tomato powder samples ranging from 0.327 to 0.413%, it is expected through synergistic reactions the desired deactivation of free radicals will be achieved leading to a reduction in oxidative stress. Essential amino acids are defined as the amino acids that cannot be synthesized by the body therefore need to be provided for in the diet for body building and maintenance of worn out tissues as modulation enzyme catalyzed pathways. Appreciable amounts of EAA ranging from 3.6 to 4.1% were found in the tomato powders which could provide the requirements for these AA by the body system for various physiological activities.
CONCLUSION
This study demonstrated that the application of pretreatments significantly affected the quality characteristics of tomato powder positively than the control samples. In all the attributes evaluated, in terms of pre-treatment the following sequence was observed K2 S2 O treated>Blanched>Control samples while the in terms of drying the following order was observed Freeze>Oven>Solar-hybrid drying. The tomato powder samples were observed to possess strong free radical scavenging activities, reduced catalytic activities and chelated transition metals, thus rendering them unavailable to promote pro-oxidant activities of free radicals and reactive oxygen species (ROS) for management of generative diseases.
REFERENCES
7. Sablani SS, Rahman MS. Pore formation in selected foods as a function of shelf temperature during freeze drying. Drying Technol. 2002; 20: 1379-1391.
8. DeLong D. How to Dry Foods. Penguin. 2006; 24-26.
16. Inc I. Spss for windows (version 22) in Chicago Ill USA. 2015.
22. Wong R, Yen G. Antioxidative activity of mungbean sprouts, soybean sprouts and radish sprouts. Chinese Agricultural Chemical Society. 1997; 35: 661-670.
31. Gordon M. The mechanism of antioxidant action in vitro. Food antioxidants. 1990; 1-18.