Folate Intake through Supplementation or Fortification - Is It Always Useful?
- 1. Department of Internal Medicine, University of Campania Luigi Vanvitelli, Naples-Italy
Abstract
Folic acid is a water soluble vitamin of B group. Folate is the generic name for B9 vitamin. This is present in some common nutriments (such as grains, cereals). Its deficiency in the diet can cause several health problems. In pregnant women, adequate folate amount is important for protect against, some congenital heart defects (CHD), neural tube defects (NTDs), or oro-facial clefts (OFC). Its serum-deficiency can be responsible for megaloblastic anemia (perniciosous), while congenital or acquired lack of folate can induce a reduced homocysteine (Hcy) serum levels, responsible for some detrimental conditions charged to cardiovascular or neurologic system and can favor an anomalous cellular replication (tumor). But, its role in the cancers’ beginning is uncertain. In fact, while some studies report that folate seems to promote the initiation and progression of certain tumours. Others refer on dual role in the malignancies, respectively in the initiation (protective) and progression and metastatization (easing). Therefore, the folate-supplementation is useful since it is optional, but foods’ fortification with folate isn’t useful always, since the growing’s enrichment with folate could be dangerous.
Keywords
• Folate
• Supplementation
• Fortification
• Neural tube defects
• Homocysteine
• Megaloblastic anaemia
• Cancers
Citation
Cacciapuoti F (2021) Folate Intake through Supplementation/Fortification - Is It Always Useful? J Hum Nutr Food Sci 9(2): 1141.
INTRODUCTION
Folic acid is the synthetic form of folate, which is its more stable compound present in vitamin B9 (also called folacin). Folate refers to the active form of the vitamin B9 and is present in some foods, such as spinach, asparagus, beef liver, eggs, dairy products, orange juice. On the contrary, folic acid is a synthetic molecule used in vitamin supplement and fortified foods. Folic acid and folate are interchangeable, that serves many functions in the human body [1].
Folic acid is converted in dihydrofolate and then in tetrahydrofolate (THF) through enzymatic reduction by the enzyme dihydrofolate reductase. Therefore, THF can be converted to the biologically active form, called L-methyl-folate by the enzyme methyilene-tetra-hydrofolate reductase (MTHFR). Vitamin B9 exerts many functions in the human body, but plays a crucial in cell growth and in the DNA formation [2,3]. Specifically, low levels of this compound are associated with an increased risk of several health conditions, such as birth defects [4], elevated homocysteine (Hcy) levels [5,6], megaloblastic anaemia [7], and carcinogenesis [8].
Folate deficiency can be due to several causes including: a diet low in fresh fruits and vegetables, some diseases that affect the folate absorption in the gastro-intestinal tract, severe kidney diseases requiring dialysis, certain medications, excessive alcohol intake, and others. The corrections of folate deficiency in the diet may be obtained by supplementation with foods/drugs or cereal and grain colture fortification [9]. Supplementation (called as dietary or nutritional supplements) consists in to add some nutrients (vitamins, minerals, some amino-acids, etc.) at diet. These supplements can be given by foods having high folate content or as drugs (pills, tablets, capsules, liquids) [10]. On the contrary, fortification consists in to add vitamins or minerals o both containing folate to cereal grain coltivations. Folate fortification can be voluntary or mandatory. Voluntary fortification is performed at the initiative of food producers to increase vitamins or minerals with high folate content in the cereals’ coltivations. This fortification type is no required by law or government regulations. On the contrary, mandatory fortification is required by law. Therefore, it is part of the government’s responsibility [11].
The normal content of folate in the body ranges between 10 and 30 ng/mL. But, its level in the serum is about 5 to 15 ng/ mL. Levels below 5 ng/mL indicate folate deficiency. Little higher content (16 to 20 ng/mL) is normally present in cerebrospinal fluid. Its metabolic products appear in the urine 6 hours after ingestion and are completely eliminated after 24 hours [12].
The unlike states of health induced by folic acid deficiency are subsequently illustrated.
Pregnancy
Folic acid is emploied by the body to make new cells and produce DNA. It is required for normal growth and development. Folate deficiency may produce some critical malformations. Among these, the most frequent are:
Neural tube defects,
Congenital heart defects,
Cleft palate,
Cleft lip
Neural tube defects (NTDs)
NTDs are a major cause of morbility and mortality among the congenital birth defects. Several epidemiological evidences seen in pregnant women indicate that folate deficiency is significantly associated with NDTs increased incidence. These are a group of birth defects that include numerous malformations, such as spina bifida, anencephaly, encephalocele, and hydrocephaly. These complex congenital impairments result from failure of neural tube closure happening during embryogenesis. Folates may be related to NDTs risk through their roles in the nucleotide synthesis. In the embryos, the rapidly dividing cells of the developing neural tube require the synthesis of nucleotids in order to facilitate DNA replication. Since folates are involved in the methylation pathway, disturbed methylation due to the folate deficiency can be responsible for the relation between folate and NTDs. But, low folate status is not a specific risk factor alone for NTDs. Some studies suggest that choline, vitamin B12, and betaine are also in [13]. Genetic and environmental factors must be esteemed too for their pathogenesis. The role of folate in NTDs formation is referred to the production of new cells; DNA and RNA, indispensable in pregnant women for induce the cytoskeleton methylation of neural cells during neural tube closure [14].
Congenital heart defects (CHDs)
Although the role of folic acid deficiency on cardiac morphogenesis remains unclear, it is evident that it attends in the migration of cardiac neural cells that contributes to the development of the heart. Folic acid may play a role in the formation of truncus arteriosus and its division in the pulmonary artery and the aorta, included the risk of transposition of great arteries and the ventricular septum formation [15].
Oro-facial clefts (OFCs)
OFCs include cleft lip with or without cleft palate. A significant protection against the occurrence of OFCs was reported in response to supplementation of folic acid. A strong association between maternal folic acid intake and the risk of develop an oro-facial deformity in babies exists. That is caused by a severe reduction in the muscle and cartilage jaw elements via DNA damage that can be induced by folic acid deficiency [16].
Homocysteine
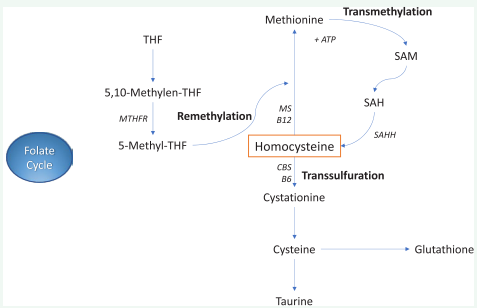
Another function of folate consists in the conversion of Hcy in Methionine, happening through the 5-methyltetrahydrofolate enzyme in Hcy cycle. Subsequently, Methionine activated by ATP, generates S-Adenosyl-Methionine (SAM), the universal methyl-donor group, that participates in numerous methylation reactions. These involve lipids, peptides, proteins, RNA, DNA and other substrates [17]. The methyl-group is transfert to these compounds through the methyltransferases enzymes. Then, SAM is turned in S-Adenosyl-Homocysteine (SAH) for -CH3 transfer. Finally, SAH is changed in Hcy + Adenosine through S-AdenosilHomocysteine Hydrolase (SAHH) (Figure 1).
Figure 1: Homocysteine cycles (Folate, Remethylation, Transmethylation, Transsulfuration).
This metabolic pathway is important for methylation of numerous substrates (lipids, proteins, DNA, RNA, neurotransmitters, etc.) and has a role for perform various cellular functions, such as chromatin remodeling, gene transcription, cell signaling and messenger RNA translation. Obviously, deficiences in folate are responsible for hypomethylation of these substrates.
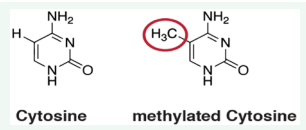
In this topic, a specific role is covered by DNA hypomethylation. This is an epigenetic mark involving the covalent transfer of a methyl group (-CH3 ) to the C-5 position of the cytosine ring of DNA (Figure 2).
Figure 2: Methylation of Cytosine of DNA.
A specific role of DNA hypomethylation is its contribution to the genomic instability and to the pathogenesis of some diseases and ageing [18].
Megaloblastic anaemia
The normal folate content in the serum represents a condition for the development of healthy red blood cells. Inversely, the unstable DNA structure generates reduced red blood cells, a condition called megaloblastic anaemia (pernicious). This is a blood disease in which the bone marrow produces large, structurally abnormal, and immature red cells (megaloblasts). Megaloblasts occur when the inhibition of DNA synthesis causes an asynchronous maturation between the nucleus and cytoplasm [19,20]. The pathophysiology of that is originated by the ineffective erythropoiesis, dependent on apoptosis of hematopoietic precursor of these cells. In turn, these abnormalities are due to thymidylate deficiency of DNA [21,22].
Malignancies
The role of folate in cancer development remains controversial and substantially unknown. On purpose, it is well known that folate mediates the transfer of one-carbon units involved in nucleotide biosynthesis and biological methylation, essential for DNA integrity. Inversely, the role of antifolates, as methotrexate, in cancer treatment is based on ineffective DNA synthesis [23]. But, the relationship between folate and cancer risk remains uncertain because some factors may influence this relation, such as the heterogeneity of the populations evaluated, the presence on polymorphism, the observations conducted pre- and postfortification, the lifestyle, and others [24]. In addition, in animal studies folate seems to have dual role in cancer development, dependent on the stage of tumor (initiation or progression). Specifically, folates’ supplementation or fortification may prevent the initiation and early promotion of cancer, these same actions may also promote the progression of cancer cells [25]. But, some animal studies have also shown that folate supplementation in these same has a protective effect on normal cells before initiation of carcinogenesis, and after their neoplastic transformation in malignancy cells, folate insufficiency inhibits tumor growth [26].
Another controversial question is if whether or not folate can prevent or promote the cancers’ development. In this regard, a number of unresolved issues there are. First, the threshold level above the folate supplementation may induce tumor-promoting effect is unknown. Furthermore, whether the tumor-promoting effect of high folate concentration is specific for synthetic folic acid alone or also acts for natural folates, as 5-methyltetrahydrofolate. [27]. Finally, the stage of neoplastic cells at which folic acid exerts a tumor-promoting effect will may be established [28]. Other concerns increasing the uncertaines about the relationship between high folate concentration and the disease include: an increased risk of bronchiolitis happening in the infant [29]. It must be also added that, contrarily to other neoplasms, leukemia [30], preeclampsia [31] and some brain tumours [32] seem to be favored by a high folate concentration.
CONCLUSION
Folate intake (supplementation or fortification) in individuals with reduced serum concentration is able to correct low plasma Hcy levels with their detrimental consequences (early atherosclerosis, Alzheimer disease, and other pathological conditions). Folate intake is also useful in to avoid some important deficiencies that can happen in early pregnancy and in to antagonize megaloblastic anaemia. But, its deficiency can obtain conflicting results on the initiation of some malignancies, on their progression and metastatic phase. So, at moment the specific mechanisms of negative effects on the different phases of malignancies are unknown. Referring to the prostate cancerinitiation, evidences from animal studies suggest that increased folate may have protective effects [33]; while others Authors indicate that decreased folate exerts protective effects [34]. Thus, further studies performed in a wide range are requested for clarify the current uncertaines of folate on the different phases of malignancies.
KEY MESSAGE
The insufficient folate amount may cause some detrimental health conditions, such as multiple birth defects, increased Hcy levels with the occurrence of their dangerous consequences (early atherosclerosis, myocardial infarction, strokes, Alzheimer or Parkinson Diseases and others), and pernicious (megaloblastic) anaemia . In these cases, its intake (with supplementation and fortification of grain and cereals’ coltivations is always useful. But, on account of the numerous uncertaines still present about the folate intake and the initiation, evolution and metastatization of some malignancies, the folate intake obtained with mandatory fortification, (as occurs in numerous States, such as US, UK, Canada, Galles, etc.) must be avoided. That on account of uncertain effects performed of folate on this tremendous pathology
REFERENCES
7. Castellanos-Sinco HB, Ramos-Penafiel CO, Santoyo-Sanchez C, CollazoJeroma J, Martinez-Murillo C, et al. Megaloblastic anaemia: folic acid and vitamin B12 metabolism. Rev Med Hosp Gen Mexico. 2015; 78: 135-143.
9. Khan KM, Jialal I. Folic acid (folate) deficiency. Treasure Island (FL). 2020.
10. Liew SC. Folic acid and diseases-supplement or not? Rev Assoc Bras. 2016; 62: 90-100.
12. Carmel R. Folic acid in: Shils M, Ahike M, Ross A, Caballero B, Cousins R. Modern nutrition in health and disease. Baltimore: Lippincott Williams & Wilkins. 2005; 470-481.
19. Pruthi RK, Tefferi A. Pernicious anaemia revised. Mayo Clin Proc. 1994; 69: 144-190.
22. Bailey LB, Gregory JF. Folate metabolism and requirements. J Nutr. 1999; 129: 779-782.
23. Shane B. Folate chemistry and metabolism. In LB. Bailey ed. Folate in health and disease. Boca Raton FL: CRC Press 2010; 1-24.
27. Smith AD, Kim YI, Refsum H. Is folic acid good for everyone? Am J Clin Nutr. 2008; 87: 517-533.
28. Kim YI. Folate and cancer: a tale of dr. Jekjll and mr. Hyde? Am J Nutr. 2018; 107: 139-142.