Investigating the Traditional Use of Silver Wafer (Chandi Ka Vark) in Inhibiting Microbial Proliferation on Sweet: A Scientific Examination of an Ancestral Practice
- 1. Department of Biosciences and Bioengineering, Indian Institute of Technology Roorkee, India
Abstract
This study investigates the effect of silver wafer (chandi ka vark) on the microbial growth and freshness of the Indian sweet, gulguley pue (dumpling puey, a simple sweet wheat dumpling which is stir fried), a culinary tradition with deep ancestral roots. Two identical samples of the sweet are be prepared: one covered with a layer of commercial-grade silver wafer and a second sample left uncovered. Both samples are be stored under normal ambient conditions (room temperature and humidity). Microbial growth is be assessed after a period of ten-weeks. Samples are be taken and examined under an upright microscope to observe and compare the morphology and presence of microbial colonies, including bacteria and fungi, on the surface of each sweet. Changes in physical appearance (color, texture, presence of mold) and sensory attributes (smell) are also be recorded. It is hypothesized that the sample of gulguley pue covered with silver wafer are exhibit less visible microbial colonization and maintain a better state of freshness compared to the uncovered control sample. The findings of this study are providing a scientific basis for understanding the potential role of silver wafer in food preservation and its impact on the shelf life of traditional sweets, while honoring the ancestral wisdom behind this practice.
Keywords
• Silver wafer
• Microbial growth
• Indian sweet
• Ancestral roots
• Investigating
Citation
Ghosh S, Tyagi S (2025) Investigating the Traditional Use of Silver Wafer (Chandi Ka Vark) in Inhibiting Microbial Proliferation on Sweet: A Scientific Examination of an Ancestral Practice. J Hum Nutr Food Sci 13(2): 1199.
INTRODUCTION
Silver, especially in its nanoscale form, has a strong toxicity towards a wide range of micro-organisms. Because of their large surface area, Ag nanoparticles also release bioactive silver ions more effectively than bulk silver. This biocidal property allows for the prevention and topical treatment of infectious diseases and also the production of antimicrobial, self-cleaning and self-disinfectant surfaces [1]. The use of silver wafer, known as chandika vark, is a long-standing tradition in many cultures, particularly in India, where it is used to adorn sweets, desserts, and even some savory dishes. This practice is not merely aesthetic but is also rooted in the belief that silver possesses medicinal and antimicrobial properties [2]. The specific sweet, gulguley pue, a dumpling-like delicacy, has been a part of Indian culinary heritage for generations, often prepared for special occasions. The application of silver wafer on such sweets is an ancestral practice passed down through families, implicitly acknowledging its role in both presentation and preservation. While its primary function appears decorative, silver has been historically recognized for its antimicrobial properties. The aim of this paper is to scientifically evaluate the potential of silver wafer to inhibit microbial growth and extend the freshness of gulguley pue. This research seeks to provide empirical evidence for a practice that is largely based on tradition and ancestral knowledge.
HYPOTHESIS
The gulguley pue sample covered with silver wafer are show delayed and less extensive microbial colonization and spoilage (e.g., mold formation, change in texture, off-odor) over a period of ten weeks compared to the uncovered control sample, providing scientific support for the ancestral practice of using silver wafer on sweets.
METHODOLOGY
Sample Preparation
- A fresh batch of gulguley pue are be prepared using a standard recipe.
- The batch are be divided into two equal and identical samples, ensuring uniformity in size, shape, and composition.
- Sample A -One sample are be carefully and completely covered with a layer of food-grade chandi ka vark (purchased from a reputable supplier to ensure purity).
- Sample B -The other sample are be left completely uncovered.
Storage Conditions
- Both samples are be placed in separate, sterile containers to prevent cross-contamination.
- The containers are be stored at a normal room temperature and humidity, reflecting typical household storage conditions. The temperature is be approximately 250C - 350C
Data Collection
- Microbiological Analysis: After a period of 10 weeks, a small, non-destructive portion of each sweet are be carefully swabbed or scraped. These samples are be mounted on a microscope slide. The slides are be immediately examined under an upright microscope at various magnifications.
- The upright microscope is particularly useful for this application as it allows for the examination of specimens, which is ideal for a solid, non transparent food sample.
- Qualitative observations of microbial growth are be recorded, including the presence, morphology and relative abundance of microorganisms. Digital images are captured to document the progression of microbial colonization over time.
- Physical and Sensory Evaluation: Observations are be recorded for:
- Appearance: Color changes, presence of visible mold, and texture.
- Odor: Presence of any off-odors.
Analysis
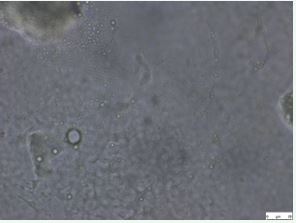
In Image 1, Sparse distribution of microbial cells with relatively fewer colonies or aggregates visible. Most cells appear small and scattered, with many individual or isolated cells rather than clusters. This image likely represents an early stage of microbial growth (lag or early exponential phase) where cells are beginning to adapt but haven’t multiplied significantly yet.
Overall surface area coverage by microbes is relatively low, suggesting limited colonization.
In Image 2, significantly higher microbial concentration. You can observe denser populations and many more visible colonies or clusters. Presence of larger aggregates or biofilm-like structures. Circular and irregular formations are evident, suggesting active cell division. This likely represents a later growth stage (mid to late exponential phase)where rapid proliferation is occurring. Much greater surface colonization and clustering, indicating advanced microbial establishment (Table 1).
Image 1 Microscope image with silver wafer.
Image 2 Microscope image without wafer.
Table 1: Summary of Differences.
|
Features |
Image with silver wafer |
Image without silver wafer |
|
Microbial Density |
Low |
High |
|
Clustering |
Mostly isolated cells |
Noticeable clusters/ colonies |
|
Surface Coverage |
Sparse |
Extensive |
|
Growth Phase Indication |
Early stage |
Mid-to-late stage |
OUTCOMES
The primary difference between the two images, using microbial growth as the basis, is the inhibitory effect of silver.
The breakdown of the differences are -
Microbial Count and Density
- Image with Silver Wafer: The number of microbial cells (the circular structures you pointed out) would be significantly lower. Silver is a well-known antimicrobial agent, and its presence would inhibit or kill bacteria, resulting in a sparse distribution of cells or even a clear “zone of inhibition” around the wafer itself.
- Image Without Silver Wafer: This image would serve as the control. There is a muchhigher density of microbial growth, with cells distributed more densely across the field of view. This represents the normal, unrestricted growth of the microbes in the absence of a bacteriostatic or bactericidal agent.
Cell Morphology and Viability
- Image with Silver Wafer: The microbes that are present show signs of damage. Silver ions disrupt key cellular processes, leading to cell wall damage, protein denaturation, and DNA damage. Under a microscope, this could manifest as irregular cell shapes, membrane blebbing, or a general loss of structural integrity.
- Image Without Silver Wafer: The cells would appear healthy and well-formed, maintaining their characteristic shapes (e.g., cocci, bacilli). Most cells would beviable, showing no signs of distress or damage.
In summary, the key difference is the impact of silver on microbial growth. The image with the silver wafer shows a suppressed and potentially damaged microbial population, while the image without the silver wafer shows a robust and flourishing population. We anticipate that the microscopic examination of the gulguley pue samples are reveal a delayed onset and slower progression of microbial colonization on the silver-covered sample compared to the control. The control sample is expected to show rapid growth of bacteria and fungi, leading to visible signs of spoilage such as mold, off-odors, and changes in texture within a few days.
The chandi ka vark-covered sample, on the other hand, is expected to remain visually and microscopically fresher for a longer period. The results of this study are expected to provide qualitative evidence of the preservation potential of silver wafer, contributing to the scientific understanding of traditional food practices and honoring the ancestral wisdom embedded within them. The Image 2 shows more advanced microbial growth compared to the Image 1. The increased density, clustering, and surface coverage indicate that microbial proliferation and colonization are much more developed in the second sample
DETAILED ANALYSIS OF CHANGES IN PHYSICAL APPEARANCE (COLOR, TEXTURE, PRESENCE OF MOLD) AND SENSORY ATTRIBUTES (SMELL) OF A SWEET PRESERVED FOR 10 WEEKS.
Color and Browning
- The initial sweet Image 3 has a uniform, dark color suggesting it’s a type of sweet like wheat along with ingredients like jaggery, all of which contribute a natural brown hue, possibly from the Mallard reaction during cooking. The final sweet Image 4 shows non-uniform color changes. The extremely dark patches could be signs of advanced oxidation or non-enzymatic browning. The lighter/ reddish-brown areas might be a result of drying and exposure.
Image 3 Sweet after making.
Texture and Hardening (Loss of Moisture)
- The most striking change is the texture. The initial piece looks soft and moist.The final pieces are clearly hardened, cracked, and shriveled, indicating a significant loss of moisture over the 10-week period. This is a common form of spoilage in confectionery, often leading to a hard, brittle, and chewy mouthfeel.
Image 4 Sweet after 10-week experiment.
Presence of Mold
- The fuzzy, patchy white/gray growth seen on the surface of the deteriorated pieces is a strong visual indicator of microbial spoilage, specifically mold growth. This is typical for sweets containing intermediate moisture levels, especially with high fat content, which can develop mold or yeast spoilage under improper storage (ambient temperature, humidity, or air exposure).
Sensory Attributes (Inferred Smell)
- Given the visible mold and the likely oxidation of fats (rancidity), the sweet would almost certainly have developed an unpleasant or “off” smell. This could include a sour, fermented, moldy, or rancid smell (due to the breakdown of fats like ghee). In conclusion, the sweet underwent significant degradation over the 10 weeks, marked by severe drying/hardening, discoloration, structural breakdown, and conclusive evidence of mold growth, rendering it spoil.
CONCLUSION
The successful execution of this experiment provides a clear and direct answer to the research question. These findings support the traditional belief that silver (chandi ka vark) contributes to the preservation of sweets. The results lead to further research on the exact mechanisms by which the silver inhibits microbial growth and could potentially have implications for the development of natural food preservation methods. Ultimately, this study aims to bridge the gap between cultural tradition and modern scientific inquiry, validating a practice that has been part of culinary heritage for centuries.
ACKNOWLEDGEMENT
The author would like to thank the Department of Biosciences and Bioengineering (BSBE), Indian Institute of Technology Roorkee, India, and Prof. Sanjoy Ghosh, Professor and Head of the Department of Biosciences and Bioengineering (BSBE), for their immense support. Authors gratefully acknowledge the constant support provided by Indian Institute of Technology Roorkee (IITR) and Ministry of Human Resource and Development (MHRD)
FUNDING
The financial assistance for this work was provided by Indian Institute of Technology Roorkee (IITR) and Ministry of Human Resource and Development (MHRD).
REFERENCES
- Schacht VJ, Neumann LV, Sandhi SK, Chen L, Henning T, Klar PJ, et al. Effects of silver nanoparticles on microbial growth dynamics. J Appl Microbiol. 2013; 114: 25-35.
- Mohamed DS, Abd El-Baky RM, Sandle T, Mandour SA, Ahmed EF. Antimicrobial Activity of Silver-Treated Bacteria against other Multi- Drug Resistant Pathogens in Their Environment. Antibiotics Basel. 2020; 9: 181.