Overview of Malting and Fermentation Role in Sorghum Flour,Primarily for Antinutrient Reduction
- 1. Department of Food science and Nutrition, Melkassa Agricultural Research, Adama, Ethiopia
Abstract
Antinutrients are phenolic compounds in crops that have a capability of binding essential nutrients into complexes and making them unavailable in both human and animal diets. Phenolic compounds, mainly phytic acid, condensed tannins, oxalate and trypsin inhibitors are common antinutrients in sorghum. Malting and Fermentation are an effective and potential biochemical process for the reduction of anti-nutritional factors in sorghum flour to safe level. Considerable amounts of phytate, condensed tannin, and oxalate and trypsin inhibitor from sorghum flour were significantly reduced as germination period elongated. The decreased antinutrients in sorghum have contributed to leaching of water soluble phenolic compounds and activated enzyme activities during malting. Reduction of tannins, oxalate, trypsin inhibitor and phytic acid content in sorghum increased with an increase in fermentation time. The reduction is as a result of exogenous and endogenous enzymes, microbial degradation and optimum pH conditions formed during fermentation. Consequently, Malted and fermented sorghum flour resulted in improved nutritive value and sensory characteristics of sorghum flour.
Keywords
- Sorghum
- Phytate
- Oxalate
- Condensed tannin
- Trypsin inhibitor
Citation
Feyera M (2021) Overview of Malting and Fermentation Role in Sorghum Flour, Primarily for Antinutrient Reduction. J Hum Nutr Food Sci 9(1): 1138.
INTRODUCTION
Sorghum is a major food and nutritional security crop to more than 100 million people in Eastern Horn of Africa [1]. It is a staple food crop on which the lives of millions of Ethiopians depend. In Ethiopia Sorghum is the third most important crop next to teff and maize in terms of cultivated area coverage and in total grain production [2]. It is a good source of energy, proteins, carbohydrates, vitamins and minerals including the trace elements, particularly iron and zinc. Sorghum grain also contains minerals such as phosphorus, Calcium, potassium and magnesium in varying quantities [3].
To be used for human consumption Sorghum is processed into a variety of traditional foods including fermented and non-fermented products such as unleavened bread, porridges, cookies, cakes, cereal extracts, malted alcoholic and non-alcoholic beverages [4]. However sorghum-based foods have continued to be nutritionally deficient and organoleptically inferior. This is largely due to the presence of anti-nutritional factors (ANF) such as tannin, phytic acid, oxalate, polyphenol and trypsin inhibitors.
Phytates are major anti-nutritional compounds identified in sorghum [5]. The phytate molecule has the capability to form insoluble complexes with proteins leading to reduced digestibility of proteins, carbohydrates and mineral bioavailability. In some extent similar to phytate, tannins from sorghum can reduce digestibility and efficiency of minerals absorption [6]. Rami found that some sorghum varieties also have protease inhibitors (trypsin, chymotrypsin, and amylase), and lectins. These antinutritional factors bind food ingredients into complexes making them unavailable for both animals and human nutrition [7,8]. In addition [9] also stated that anti-nutritional factors in sorghum limit the digestibility of both proteins and carbohydrates by inhibiting their respective proteolytic and amylolytic enzymes. According to Ra [10] those antinutrient compounds are more diverse in nature and are present in higher amount in sorghum than in other cereal crops like wheat, barley, rice, maize, rye, and oats. Sorghum has been processed into different traditional food products in Ethiopia so far. still processed food from sorghum is nutritionally less available and organoleptically inferior. This is due to the presence of antinutrients such as Tannin, phytic acid or phytate, oxalate, polyphenol and trypsin inhibitors in sorghum. Those antinutrients have a capability of binding nutrient ingredients into complexes and making them unavailable for animals and human nutrition [7,8]. Processing techniques such as malting and fermentation have positive contribution not only to reduce antinutrient contents of grain but also enhance nutrient composition, functional properties, and organoleptic qualities. According to research reported by [11] Malting and fermentation can have multiple effects on the nutritional value of food.
Fermentation has been reported as an effective biochemical process in the reduction of anti-nutritional factors in cereals and improves their starch and protein digestibility, amino acid balance as well as nutritive value [12]. As reported by [13], Malting of cereal caused an improvement in protein digestibility and other protein quality characteristics, such as percentage of protein, nitrogen solubility index and content of the first limiting amino acid, lysine with increasing time. According to [14] combination of germination followed by the fermentation changes major biochemical component when compared to germination alone, so these processes is a potential for developing of the improved nutritive value of food product with high nutrients and minerals availability.
Germination or malting is a biochemical process which involves the transition of a seed from a dormant state to a vital active state. The primary aim of malting is to promote the development of hydrolytic enzymes, which are not present in non-germinated grain [13]. Malting caused an improvement in protein digestibility and other protein quality characteristics, such as percentage of protein, nitrogen solubility index and content of the first limiting amino acid, lysine with increasing time [13], physical and biochemical factors such as temperature, time of steeping and germinating of grains with their inherent enzymic activities, kilning temperature influence quality of malted sorghum flour [15].
Fermentation is another important process which significantly lowers the content of anti-nutrients (phytates, tannins, polyphenol) of cereal grains [16]. Fermentation also provides optimum pH conditions for enzymatic degradation of phytate which is present in cereals in the form of complexes with polyvalent cations such as iron, zinc, calcium, magnesium, and proteins. Such a reduction in phytate may increase the amount of soluble iron, zinc, calcium several folds [17]. Combination of germination followed by fermentation changes major biochemical components when compared to germination alone, so these processes is a potential for developing food production of improved nutritive value with high minerals availability [14]. Total polyphenols, tannins and phytates in sorghum-pumpkin composite flour varied with fermentation time ranging from 0-36 hours [18].Therefore, the objective of this article is to review the effect of malting and fermentation on antinutrients in sorghum flour.
SORGHUM PRODUCTION AND UTILIZATION
Sorghum is a major cereal crop in arid and semi-arid areas of the world. It is a cereal native to sub-Saharan Africa and grows well in temperate and tropical areas of the world where other staple cereals such as maize, wheat, and rice cannot grow well [19]. In 2014 World annual crop production report indicate that sorghum production is over 60 million tonnes, of which Africa produces about 20 million tones [20]. This makes sorghum, quantitatively the second most important cereal grain in Africa after maize. Nigeria, Sudan, Ethiopia, and Burkina Faso account for nearly 70% of Africa’s sorghum production [21].
According [1] Sorghum is a major food and nutritional security crop to more than 100 million people in the Eastern Horn of Africa. In Ethiopia Sorghum is the third most important crop next to teff and maize in terms of cultivated area coverage and in total grain production [2]. It is a staple food crop on which the lives of millions of Ethiopians depend. Currently, sorghum is produced by nearly 5 million holders and its production is about 43 million quintals (Qt) from around 2 million hectares of land giving grain yield of 23 Qt per hectare [2]. Sorghum cultivated in all regions of Ethiopia between 400m to 2500m altitude. Oromiya, Amhara, Tigray and Southern nations, nationalities and people representatives (SNNPR) are a major producer of sorghum in Ethiopia.
Sorghum has a wide variety of uses [22]. The grain is eaten after boiling the flour to produce foods such as ugali, sadza, and Uji. In Ethiopia, sorghum flour is used to make injera, traditional bread made from fermented dough. Sorghum grain is also used for brewing. Varieties of sorghum suitable for brewing have low tannin content since consumers prefer beer with this taste. Although sorghum grain is not usually fed to livestock, sorghum Stover is used for fodder as well as fuel and material for building and roofing houses.
NUTRIENT COMPOSITION OF SORGHUM
Sorghum grain is the potential source of starch, proteins, lipids, non-starch polysaccharides and phytochemicals such as phenolic compounds, phytosterols, and policosanols. Sorghum grain also contains dietary fiber, including resistant starch, and micronutrients including vitamins and minerals, oil bodies and waxes. Sorghum grain is a good source of starch, containing approximately 71% of dry whole grain weight [23]. The starch is encapsulated in granules that are located predominantly in the endosperm (storage tissue), though uniquely some are present in the pericarp outer layer of grain [23].
Sorghum starch is comprised of both amylose (linear polymers of glucose) and amylopectin polysaccharides (branched polymers of glucose). Several research report indicate that very low percentages of amylose present in the starch of waxy sorghum varieties compared to 24-33% in non-waxy sorghum starch [24,25] Duodu report that Sorghum starch granules are enmeshed in a strong protein matrix in the endosperm, a unique structural aspect of sorghum grain. Disulphide-bond cross-linking involving kafirins in the protein matrix forms a protective network around the starch granules reducing starch digestibility [26]. The lower starch digestibility reported for sorghum foods is not an intrinsic property of the sorghum starch granules themselves, but appears mainly to be a consequence of the interactions of the starch with the endosperm protein matrix, as well as with cell wall material and polyphenolic compounds, such as condensed tannins and flavonoids [27].
Protein is the second largest constituent of sorghum grain (6- 18%) after starch [28]. Sorghum endosperm proteins are found in both a matrix and as protein bodies that are enveloped by the matrix. Sorghum proteins are classified as albumins, globulins, kafirins, cross-linked kafirins and glutelins [29]. Of these, kafirins are the main protein [29], comprising 50-70% of the total protein content [25]. The kafirins are prolamin storage proteins with limiting levels of some amino acids, in particular, lysine. The kafirins differ in structure from the gliadin and glutenin storage proteins in wheat. They do not elicit damage to the mucosa of the small intestine of people with coeliac disease making sorghum a viable ingredient for gluten-free foods such as bread.
Sorghum grain contains approximately 3-4% lipids, the majority of which are neutral triglycerides, rich in unsaturated fatty acids and mostly present in the germ [30]. The predominant fatty acids are oleic acid (31.1–48.9%), linoleic acids (27.6– 50.7%), linolenic acid (1.7–3.9%), stearic acid (1.1–2.6%), palmitic acid (11.7–20.2%) and palmitoleic acid (0.4– 0.6%) [31]. Two less common saturated fatty acids, octanedioic (C8:0) and azelaic acid (C9:0), have been identified in some sorghum varieties [31]. This lipid composition has generated interest in sorghum as a source of edible oil, representing a potentially valuable dietary source of monounsaturated fatty acids (MUFA) and polyunsaturated fatty acids (PUFA), with higher PUFA levels than MUFA [31]. This desirable lipid composition is conducive to mechanisms that lower lipid levels in humans and therefore to potentially lower risk factors associated with heart disease. Sorghum also contains plant sterols that may reduce cholesterol absorption to collectively lower plasma and liver cholesterol concentrations [32]. These results were recently supported by [33]. In a hamster model of hypercholesterolemia, investigating the effects of whole kernel grain sorghum oil (rich in plant sterols) and wax (high in policosanols).
As with cereals more broadly, sorghum is a source of B-complex vitamins (such as thiamin, riboflavin, vitamin B6, biotin, and niacin) that are diminished with grain refining processes including decortication [34]. The mineral composition in sorghum is similar to millet, higher compared to maize but lowers than wheat, and is predominantly composed of potassium and phosphorus (34. Sorghum-based foods are a good source of both iron and zinc, although anti-nutrients such as phytates may diminish bioavailability [27]. A problem not unique to sorghum but common to other grains and plant foods in general.
HEALTH BENEFITS OF SORGHUM
Sorghum may be a valuable lower-calorie grain alternative in Western diets where overweight and obesity rates continue to rise and represent major public health burdens (WHO, 2010). Sorghum’s energy value is approximately 1377 kJ/100g; however, the available energy for human metabolism may be lower than this estimate due to the described low starch and protein digestibility rates. This postulation is partly based on evidence from numerous feeding studies that show animals (from rodents to livestock species) fed whole grain sorghum, in particular, the slowly digested high tannin sorghum varieties, have reduced weight gain [35]. In general, dietary fiber and whole grain intakes have been associated with reduced risks of obesity, overweight and with the lowered waist-to-hip ratio [36]. Most of the research identifying beneficial effects of sorghum consumption still lies with animal models [37]. Epidemiological evidence dating back to the early 1980s has correlated consumption of sorghum with reduced incidence of esophageal cancer, warranting closer attention to the potential chemo preventive properties of sorghum chemical components. Data from various sorghumconsuming countries in Africa and Asia have demonstrated lower esophageal cancer incidences compared to regions where wheat and maize were the major cereals consumed [38].
FUNCTIONAL PROPERTIES OF SORGHUM
Functional properties are those parameters that determine the application and end use of food materials for various food products. The application of flour or starch in food production and in the industry depend on various functional properties such as dispersibility, water absorption capacity, pasting, retrogradation, viscosity, swelling power, solubility index etc. which varies considerably based on the type of crop as well as an ecological and agronomic influence [39]. These functional properties depend on the composition and molecular structures of the flour or starch which include amylose/amylopectin ratio, phosphorus content, starch molecular weight, granule size and the chain length distribution [40]. Bulk density is an important parameter for determining the easy ability of packaging and transportation of particulate or powdery foods. Dispersibility is a measure of reconstitution of flour or starch in water, the higher the dispersibility, the better the sample reconstitutes in water and gives a fine constituent during mixing [41].
Swelling power indicates the water holding capacity of floury samples, which has generally been used to demonstrate the differences between various types of flours. [42] Jasmien found that the lower the starch content, the lower the swelling power. This may be due to the hydrophilic nature of starch which enhanced water uptake as the starch content increases enhancing swelling occurrence. Water solubility index determines the number of polysaccharides or polysaccharide release from the granule on the addition of the excess of water. Water absorption capacity or characteristics represent the ability of a product to associate with water under conditions where water is limited [12]. WHC gives an indication of the amount of water available for gelatinization and low absorption capacity is desirable for making thinner gruels as reported by [12]. Fermentation and malting can improve the functional properties of grain flour. Research report shows that there is a significant increase in the water holding capacity of germinated and fermented sorghum flour. Water absorption of the flour mainly depends upon protein, carbohydrate, their interaction and nature [43].
MALTING OF SORGHUM
Malting is the controlled germination followed by controlled drying of the kernels. The main objective of malting is to promote the development of hydrolytic enzymes, which are not present in non-germinated grain [13]. Sorghum in vitro digestion studies shows that malting caused an improvement in protein digestibility and other protein quality characteristics, including percentage of protein, nitrogen solubility index and content of the first limiting amino acid, lysine [13]. Other benefits of the malting process include increased vitamin C content, phosphorus availability, and synthesis of lysine and tryptophan. Also during malting, both starch and protein are partially degraded allowing for better digestibility. Furthermore amylases are elaborated and as a result, the viscosity of gelled starch decreases.
Malting has produced improvement in flavor profile and color [44]. Research conducted on the improvement of the protein quality of sorghum and its introduction into staple food products for southern and eastern Africa showed that malting, in addition to improving the malt quality characteristics, also improved the digestibility and quality of the protein, which generally increased with increased malting time .Malting has the potential for antinutrient reduction in various grains.
According to [8], Activity of phytase enzyme, reduce of phytate is by degrading of phytate into inorganic phosphorus and myo inositol and its intermediate forms. The higher the sprouting time the more effective the reduction of oxalate [45]. Therefore, in view of these multiple benefits, use of a malting processing method needs to be advocated. The process of malting comprises three unit operations: steeping, germination, and drying. During steeping, kernels are immersed in water until imbibed with sufficient water to start the metabolic processes of germination and at the same time dirt, chaff and broken kernels are removed by washing and flotation. The germination phase begins after the kernels have absorbed enough water to start enzyme production and starch hydrolysis.
Conditions that are necessary during the germination phase are moisture content, temperature, length of germination time, and oxygen availability. Germination takes about 4-6 days and occurs rapidly between 20°C and 30°C with an optimum temperature of 25°C to 28°C [46]. The most important physiological processes associated with the germination phase are the synthesis of amylases, proteases and other endogenous hydrolytic enzymes [46] during the process, the hydrolytic enzymes migrate from the germ into the endosperm where starch and protein are hydrolyzed to sugars and amino acids, respectively. These are then transported into the germ where they are further metabolized by the growing seedling [46].
Drying is the final stage of the malting process and is required for stopping further growth of the kernels, reducing the moisture content and water activity, hence producing a shelf-stable product with active enzymes [46] Kernels are dried at a temperature of about 50°C for 24 hours [46]. After drying the roots and shoots are removed and the kernels milled into malted flour ready for use in the preparations of different food products.
FERMENTATION OF SORGHUM
Fermentation can be defined as a desirable process of biochemical modification of primary food matrix brought about by microorganisms and their enzymes. Fermentation of cereals leads to a general improvement in the shelf life, texture, taste and aroma, nutritional value and digestibility and significantly lowers the content of antinutrients of cereal products. Its importance in modern-day life is underlined by the wide spectrum of fermented foods marketed both in developing and industrialized countries, not only for the benefit of preservation and safety but also for their highly appreciated sensory attributes [47].
Several studies have been showed that fermentation is effective techniques in decreasing the level of antinutritional factors in cereals and improves their starch and protein digestibility, amino acid balance as well as nutritive value [12]. Fermentation caused an increase in the amounts of soluble proteins and the free amino acids in sorghum flour. The increase can be attributed to the microbial synthesis of proteins from metabolic intermediates during their growth circles. Fermentation also increases vitamin B content particularly thiamine and improve mineral availability [48]. It can improve nutrient density and increase the amount and bioavailability of nutrients through degradation of antinutritional factors, pre-digestion of certain food components, synthesis of compounds that improve absorption and by influencing the uptake of nutrients in the intestine [49].
Fermentation can increase in the concentrations of vitamins, minerals, and protein. Fermentation can also improve mineral availability and increase vitamin B content particularly thiamine [48]. Using different combinations of starter organisms fermentation reduces polyphenol, tannins and phytic acid content with an increase in fermentation time [50]. The reduction is as a result of exogenous and endogenous enzymes such as phytase enzyme formed during fermentation. Fermentation also provides optimum pH conditions for enzymatic degradation of phytate which is present in cereals in the form of complexes with polyvalent cations such as iron, zinc, calcium, magnesium, and proteins. Such a reduction in phytate may increase the amount of soluble iron, zinc, calcium several folds [51]. During fermentation, the reduction of phytic acid content may partly be due to phytase activity as it is known to be possessed by a wide range of microflora. The optimal temperature for phytase activity has been known to range between 35°C and 45°C [16]. Tanin levels may be reduced as a result of lactic acid fermentation, leading to increased absorption of iron, except in some high tannin cereals, where little or no improvement in iron availability has been observed [51]. Diminishing effect of fermentation on polyphenols may be due to the activity of polyphenol oxidase present in the food grain or microflora [16].
PRODUCTION OF MALTED AND FERMENTED SORGHUM FLOUR
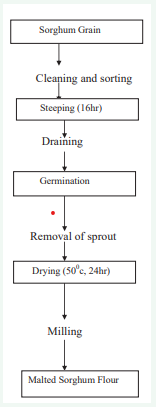
Malting could be done according to the method described by [52], with a slight modification as shown in Figure 1.
Figure 1: Flow diagram for Malted sorghum flour production.
Sorghum grains was sorted and cleaned manually to remove foreign matter, soaked for 16 h in tap water (w/v; 1:2). Soaked grains were drained and sprouted by spreading out on a covered jute bag. Water then, sprinkled on it daily until sprouting began. Sorghum grains were subjected to different sporulating duration. After germinating, germinated sorghum kernel was dried in an oven at a temperature of about 50°C for 24 hours [46]. After drying the roots and shoots rubbed by hand and removed using manual winnowing. Then the dried kernels milled into malted flour.
The fermentation carried out according to, whole Sorghum flour mixed with water (distilled) in the ratio of 1:2 (w/v) to make dough and allowed to ferment at room temperature for different fermentation time in a cleaned plastic container covered with plastic material. The dough will be fermented by adding 5% starter culture from previously fermented dough to start the fermentation process. The fermented dough will be dried in laboratory oven dry at 700 c for 16 hr. The dried samples then milled to a fine powder using a laboratory miller machine that passed through a 0.5 mm sieve size (Figure 1).
Utilization of Malted and Fermented Sorghum in Food and Beverages
Sorghum, like other cereals, is an excellent source of starch and protein and can be processed into starch, flour, grits and flakes which can be used to produce a wide range of food, feed, and industrial products. Malted and fermented sorghum flour can be processed into malted foods, beverages, and beer. Cakes, cookies, pasta, a parboiled rice-like product and snack foods have been successfully produced from sorghum. Porridges appear to be the most common types of food prepared from sorghum. A range of porridges of varying consistencies (soft or thick) maybe prepared from fermented or non-fermented sorghum meal. Flatbread and alcoholic beverages are also produced from malted fermented sorghum. Porridges made from composites of extruded plain/malted/fermented sorghum flours seem to offer the best route to address of child mal nutrition. The malted fermented sorghum flour also resulted in improved sensory characteristics of cowpea-cassava danwake samples. The effects of malted fermented sorghum on the quality characteristics of cowpea-cassava danwake were investigated as technological strategies for improving the quality of danwake flour. Fermented sorghum flour has many benefits in the bread products, improving texture and mouth-feel, acceptability and shelf-life of bread. Composite flour produced from malted sorghum flour with 10% soy flour substitution could be used to produce organoleptically acceptable product.
ANTINUTRIENTS IN FERMENTED AND MALTED SORGHUM FLOUR
The nutritional profile of sorghum can be compromised to a certain extent by its content and activity of antinutrients (AN): phenolic compounds, mainly condensed tannins, oxalate, trypsin inhibitor and phytic acid. All anti nutrients interact negatively with the bio accessibility of essential elements in the digestive tract particularly iron and zinc; moreover, tannins further reduce the digestibility of sorghum’ proteins. The general mechanism involves the formation of insoluble complexes at physiological pH, due to the ability of phytic acid and tannins to bind proteins and divalent cations. The tannins are also able to bind human gut enzymes involved in the cereal digestion. As a consequence, the AN elicit an unbalanced intake of essential elements as well as reduce the availability of metabolizable energy and amino acids. Similarly, the presence of high levels of tannins in cereals, such as sorghum, can result in significantly reduced protein and amino acid digestibility [53]. However, they could be eliminated or reduced by processes such as soaking, dehulling, germination and fermentation [54,55]
Phytic acid (PA)
phytic acid is the primary storage compound of phosphorus in cereals, legumes, nuts, and oilseeds. It accounts for up to 90% of total phosphorous content and contributes as much as 1.5% to the seed dry weight [56]. Its principal functions in seeds are the storage of phosphates as a source of energy and the antioxidant activity of the germinating seed [57]. The amount of PA in plants is very variable and, presumably, it depends on growing conditions and harvesting techniques [58]. Nevertheless, phosphorus in PA is mostly not bioavailable to monogastric animals, including humans, due to insufficient degradation capabilities in their gastrointestinal tract under the pH conditions of the small intestine [58]. Like tannins, also PA has the ability to chelate metal cations, primarily iron, zinc, calcium, as well as proteins and digestive enzymes, such as pepsin, amylase, and trypsin. The formation of insoluble complexes with metals and proteins determines their unavailability as nutrients and can lead to deficiencies in populations where staple foods like sorghum represent the principal source of nutrition. In Africa, sorghum is frequently used for preparing weaning food for children: the intake of phytates has a direct correlation with the poor iron and zinc status commonly seen in weaned, preschool children after 6 months of age in low-income countries, e.g., Malawi [59]. Additional studies have shown the PA ability to inhibit the absorption of further essential minerals, including calcium and magnesium [60]. In particular, the PA-induced impaired bio accessibility of zinc may be of particular relevance for the well-being of the developing organism [61]. Fermentation is biochemical reaction capable of creating acidity (low PH) medium in fermented ingredient and caused greater reduction in phytic acid. The optimal PH for cereal phytase is around 5.0, according to research finding by [62]. phytase enzyme activity is very high during fermentation process. Similar research revealed that at low PH, microbial phytase activity during fermentation accounts significant reduction in phytic acid in the fermented product through hydrolyses of phytic acid to inorganic phosphate and inositol [63-65]. Such a reduction in phytate may increase the amount of soluble iron, zinc, calcium several folds [17]. Several seeds or grains are known to contain phytase enzyme and its activity varies widely. Phytase activity increased during the germination of several seeds [66]. The principal function of phytase in seeds or grains is to liberate phosphate from phytate during germination. Germination reduces and/or eliminates considerable amounts of phytate from the seeds or grains. [66] Valencia reported a significant reduction of (phosphate) of 38% and 35% in quinoa seed samples. Sprouting of Kaura sorghum for 48 hours reduced (80.29%) of phytate [67]. Maidala also reported that phytic acid reduction in sorghum increased with higher the sprouting time. According to research conducted by [68] on sorghum grist subjected to different steeping and germination period revealed that Phytate levels were significantly reduced from 1.50% in sorghum grist to 0.51% in 48 hour steeped and 96 hour germinated sorghum grist. During malting and fermentation endogenous enzymes such as phytases have been activated. Research finding reported by [65] revealed that significant reduction of phytic acid from sorghum cultivars were associated to degradation of phytate into inorganic phosphorus and inositol and its intermediate forms by Phytases enzyme activity. According to [69] malting and fermentation reduced phytate content of sorghum flour significantly up to 40% and 77%, respectively. Fermentation is the most effective processing technique that reduced phytic acid in the cereal flours [70]
Condensed Tannin
Tannins are water–soluble phenolic compounds that have the ability to bind or precipitate proteins from aqueous solutions. They bind with storage proteins of sorghum and inhibit their digestibility. The anti-nutritional effect of tannins includes diminished growth rate, the bioavailability of mineral elements and decreased feed efficiency in rats, swine, poultry, and ruminants. The tannins reduced the availability of minerals, proteins, and starch of the sorghum [71] Interaction of tannins and other sorghum phenolic compounds with starch and effects on in vitro starch digestibility. Tannins from sorghum reduce digestibility and efficiency of mineral absorption. The presence of condensed tannins in sorghum cultivars also greatly affects the functional and nutritional quality of sorghum food products. it reduce protein digestibility of sorghum [72] because they bind with proteins and inhibit enzymes [74]. Fermentation is an important process which significantly lowers the content of condensed tannins in cereal grains [16]. Condensed Tanin levels may be reduced as a result of lactic acid fermentation, leading to increased absorption of iron, except in some high tanin cereals [74]. Fermentation reduced the condensed tannin content of sorghum flour from 3.1 mg/g for unfermented one to 0.1 mg/g, for 24 hour fermented sorghum flour [69]. The tannin levels in sorghum grist subjected to different germination period were significantly reduced with increased steeping and germination periods [68].
[69] Ojha also reported reduction of the condensed tannin from 3.1 mg/g for unprocessed sorghum flour to 2.6 mg/g for sorghum flour steeped for 24 hour and germinated for 48 hour. Other related research conducted by [67] showed that Malting of Kaura sorghum for 48, 78 and 92 hours reduced 96.33%,96.48% , 96.48% of tannin content. This finding indicated that as the sprouting time increased the efficiency of tannin reduction is increased. Water used during steeping step caused leaching of water soluble tannin and have contributed in the reduction of condensed tannin and some of the other anti-nutritional factors. Several studies have reported that decreased in tannins content in cereals were observed after subjected to different fermentation and germination process [75-77]. Red sorghum contains the highest amount of tannin and provides more resistance to tannin reduction as a result of malting. Reduction in tannin is brought about by leaching as reported by [54,55].
Oxalate
Oxalates also known as oxalic acid is a naturally occurring compound in plants. These plant-based oxalates are consumed through our diet as well as produced as waste by our bodies. The oxalate content of food can vary considerably between crops even of the same species, due to differences in Agro climate, soil type, stage of ripeness, or even which part of the plant is analyzed. Variations also may be caused by the different methods used for measuring oxalate in food. Various foods high in oxalates are rich in many nutrients beneficial to your health, like leafy greens and legumes. However, because oxalates bind to calcium as they leave the body, they can increase the risk of kidney stones in some people. Several processing methods such as germination, fermentation, and boiling were found to reduce oxalate content to save level 80 mg/g [78]. The mean of the oxalates content was reduced significantly in sorghum and pumpkin fermented with Aspergillus niger at room temperature from 1.69mg/100g for unfermented sample to 0.23mg/100g for 72 hours fermented samples [79]. Related research report on malting of Kaura sorghum for 48 hours showed significant reduction (86.67%) of oxalate [67].
Trypsin inhibitor
Trypsin inhibitors are phenolic compounds widely spread in legumes and cereals. They inhibit proteolytic enzymes in digestive system [80] and cause pancreatic hypertrophy and poor growth performance [81]. Different processing methods such as germination, fermentation, thermal and radiation were found to reduce trypsin inhibitor to save level and enhance protein and starch digestibility [62,82]. Significantly reduced trypsin inhibitor activity level in the three sorghum cultivars were noted 87.4% in ahmer, 77.7% in Faterita and 76.5% in Safra after 24 h fermentation [83]. Similar results found by [83] were also indicated appreciable reduction (32%) in trypsin inhibitor of corn meal fermented at 32ºC for four days. The reduction in trypsin inhibitor activity could be attributed to microbial degradation of trypsin inhibitor during lactic acid fermentation. According to [68] trypsin inhibitor activity in sorghum grist subjected to different steeping and germination period was reduced by 36.5%.
CONCLUSION
Sorghum is a staple and nutritional security crop that is vital to provide energy, proteins, carbohydrates, vitamins and minerals to more than 100 million people in Eastern Horn of Africa. Anti-nutritional factors in sorghum limit the digestibility of both proteins and carbohydrates by inhibiting their respective proteolytic and amylolytic enzymes. Bio chemical processing such as germination and fermentation were effective to reduce antinutritional factors save level. High percentage of phytate, condensed tannin, trypsin inhibitor and oxalate reduction were observed at elongated malting time 92 hours and fermentation time upto 96 hours. Optimally, malted and fermented sorghum flour could be, used in production of nutritious food alone or through blending with legumes, fruits, vegetables, root and tubers.
REFERENCES
4. Roone LW and Serna-Saldivar, SO Sorghum. In: Handbook of Cereal Science and Technology. New York.
11. Brigas, DE; Hough, R; Young,TN. Malting and Brewing Science Malt and Sweet wort. 2nd Edn.Chapman and Hall, UK.p.295-296.
15. Pathirana RA. Shivayogsundaram K and Jayatissa PM. Optimisation of conditions of malting of sorghum. J. Food Sci. Technol. 1983; 20: 108.
16. Sindhu SC, Khetarpaul N. Probiotic fermentation of indigenous food mixture: Effect on antinutrients and digestibility of starch and protein. J. Food Compos. Anal. 2001; 14: 601-609.
20. FAOSTAT. FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS. Retrieved August 5, 2014. 20.
21. FAO. Annual Publication. Rome, Italy: Food and Agricultural Organization.
22. Serna-Saldivar S, Rooney LW. Structure and chemistry of sorghum and millets. In:Sorghum and Millets Chemistry and Technology. AACC; 1995; 69: 123.
29. Glew RH, Vanderjagt DJ, Lockett C, et al. Amino Acid, Fatty Acid, and Mineral Composition of 24 Indigenous Plants of Burkina Faso. J Food Compost Anal. 1997; 10: 205.
33. Hegedüs, M.; Pedersen, B.; Eggum, B.O. The influence of milling on the nutritive value of flour from cereal grains. 7. Vitamins and tryptophan. Qualitas Plantarum 1985: 35: 175-180.
43. Rooney LW and Waniska R.D. Sorghum food and industrial utilization. pp.689-729. In: C.W. Smith and R.A. Frederkisen, (eds). ‘Sorghhum: Origin, History, Technology, and Production, John Wiley and Sons, New York.
48. Leder I. Sorghum and millet in cultivated plants, primarily as food sources. UK: UNESCO, EOLSS, Publisher Oxford UK.
49. Wakil SM & Kazeem M O. Quality assessment of weaning food produced from fermented cereal-legume blends using starters. Int Food Res J. 2012; 19: 1679-1685.
51. IITA Cereal in Tropical Africa. A Reference Manual, International Institute of Tropical Agriculture.1990; Ibadan, Nigeria. 7-8.
56. Raboy V. Myo-Inositol-1,2,3,4,5,6-hexakisphosphate. Phytochemistry. 2003; 64: 1033–1043.