Intentional Overdose with Insulin Glargine in a Non Diabetic Patient
- 1. Faculté de Médecine de Tunis, Université Tunis El Manar, Tunisie
Abstract
Background: Suicide attempts by injecting insulin glargine in non-diabetic patients are rare. Few cases have been reported.
Case report: A 49 year old man with a history of non treated disorder behavior invokes emergencies 3 hours after injecting 500 units of insulin glargine (lantus®). He was asymptomatic with normal blood glucose finger level (90mg/ml). The first episode of hypoglycemia (50mg/dl) occurred six hours after injection.
He was then transferred to the intensive care unit.He was treated with continuous intravenous infusion of 1700 g of carbohydrate with potassium supply by central venous line for severe hypokalemia at 2.8 meq/l. The last episode occurred 57 hours after the overdose insulin injection.
Conclusion: Prolonged hypoglycemia and hypokalemia are two fatal complications of poisoning with insulin glargine requiring close monitoring in the intensive care unit. This monitoring should be prolonged to avoid early interruption of dextrose infusion.
Keywords
• Insulin glargine
• Hypoglycemia
• Suicide attempt
• Hypokalemia
Citation
Fatma E, Aymen MR, Youssef B, Nozha B (2017) Intentional Overdose with Insulin Glargine in a Non Diabetic Patient. J Pharmacol Clin Toxicol 5(4):1081.
ABBREVIATIONS
H: Hour
INTRODUCTION
Suicide attempts by injecting insulin glargine in non- diabetic patients are rare. Some cases have been reported to focus on the high risk of prolonged hypoglycemia [1-9].
CASE PRESENTATION
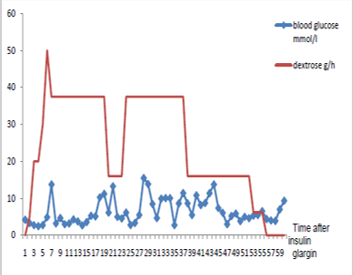
We report a 49-year-old, non-diabetic, 100-kg male, suffering from behavioral disorder without treatment who invokes emergencies 3 hours after injecting his mother’s insulin glargine (lantus®) in a suicide attempt. He injected 500 units in different sites: left arm, both thighs. The initial clinical examination revealed an asymptomatic patient with a blood glucose finger of 90 mg/dl. The laboratory tests showed hypokalemia with serum potassium 2.8 meq/l (normal: 3.5-4.8 meq/l), sodium 143 meq/l, glucose 4.2 mmol/l, urea 3.2 mmol/l and creatinine 56 μmol/l. His electrocardiogram was normal. A continuous infusion of 5% glucose started at a rate of 100 ml/h (i.e. 5g/h). Six hours after injection, the patient presented hypoglycemia (50 mg/dl) without any clinical manifestations. He was then transferred to intensive care unit. The blood glucose level was monitored hourly with glucometer (Figure 1),
Figure 1 Evolution of finger stick glucose.
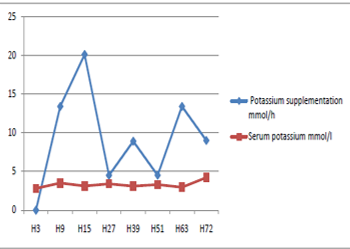
and serum potassium was measured twice a day (Figure 2).
Figure 2 Serum potassium versus potassium intake intravenously.
He was treated with continuous intravenous infusion of 30% dextrose at an initial rate of 125 ml/h (i.e. 37.5 g/h) (Figure 1), high caloric oral feeding and potassium supply by central venous way. The rate of glucose infusion was progressively decreased and was discontinued 67 hours after injecting insulin glargine. During the course of hospitalization, nine episodes of hypoglycemia with tremor were recorded requiring treatment with bolus of 30 g of dextrose. The last episode was detected 57 hours after the overdose insulin injection. The patient recovered and he was discharged to home 73 hours after poisoning.
DISCUSSION
Insulin glargine is the first long-acting insulin analogue used for basal insulin therapy to manage diabetes. It’s administered subcutaneously once daily at bedtime and released very slowly into the blood plasma over 24 h [10,11].
The major risk of poisoning is profound hypoglycemia which can induce severe neurological damage [6]. Given the long-acting nature of insulin glargine, the poisoned patients need close monitoring even in the absence of serious symptoms. According to our knowledge, eleven cases of overdose were reported in the literature (Table 1).
Table 1: Summary of published cases of poisoning with insulin glargine.
| Age/sex (reference) |
33/ |
21/ |
22/ |
31/ |
37/ |
51/ |
12/ |
39/ |
56/ |
55/ |
46/ |
49/M (our patient) |
| Diabetes | no | yes | yes | no | yes | yes | no | yes | yes | yes | yes | no |
| Consultation period (h) | 5 | 2,5 | 4h | 3 | 5 | 16 | 1-2h | - | 1-1,5 | 20 min | 15 min | 5 |
| Intentional poisoning | yes | yes | yes | yes | yes | yes | yes | yes | yes | no | no | yes |
| Site of injection | multiple | 1 | 2 | multiple | multiple | multiple | 1 | multiple | 1 | 1 | 1 | multiple |
| Isulin glargine dose | 300 | 26 | 300 | 1000 | 150 | 2700 | 2000 | 3800 | 3300 | 100 | 100 | 500 |
| Other insulin (type) | yes (as-part) | no | yes (as-part) | no | no | no | no | yes (lis-pro) | no | no | no | no |
| Lowest blood glucose reported(mg/ dl) | 38 | 16 | undetec table | 43 | 15 | 23 | 25 | 50 | 30 | 134 | 134 | 45 |
| First episode of hypoglycemia (h | <8 | 2.5 | 4 | 3 | 5 | 16 | 2 | - | 1.5 | 6 | ||
| Last episode of hypoglycemia (h | 14 | 53 | 30 | 106 | <24 | 120 | 63 | - | 72 | - | - | 57 |
| Duration of glucose infusion(h) | 40 | 60 | 59 | 130 | >48 | 120 | 130 | 108 | >100 | - | - | 67 |
| Serum potassium | - | 3.3 | - | 3.4 | 3.3 | 3.3 | 3 | - | - | - | - | 2.8 |
Most of them (n=8) occurred in diabetics [2,3,5,6,8,9,12,13]. Two patients, with unintentional overdose, were observed for 17 and 24 h respectively and they didn’t exhibit hypoglycemia [12,13]. The nine intentional cases [1-9] experienced signs of hypoglycemia within 1.5 to 16 h after injecting subcutaneously 26 to 3800 units of insulin glargine associated to insulin aspart in two cases [1,3] and insulin lispro in one case [8]. They were treated by continuous glucose infusion for 40 to 130h and they presented the last episode of hypoglycemia over 14 to 120 h after overdose [1-9]. In association with dextrose infusion, other treatment modalities such as glucagon [14,15], octreotide [9] and incision of the injection site [5,15,16] was proposed in insulin overdose. Only octreotide was tried successfully in insulin glargine [9]. Our patient was initially asymptomatic and the first episode of hypoglycemia occurred 6 hours after poisoning. He required 1700 g of dextrose administered intravenously in association with oral carbohydrate intakes during 67 hours. He developed hypoglycemia for the last time 57 h after the overdose.
Hypokalemia is the other complication associated with the use of all insulins types particularly in case of poisoning. It was reported in 5 cases with serum potassium between 3 and 3.4 meq/l [2,4,5,7,8]. Our patient had severe hypokalemia (2.8 meq/l) requiring supplementation by central line venous.
CONCLUSION
Although poisoning with insulin glargine is relatively rare, it can be dangerous for the risk of deep and prolonged hypoglycemia and severe hypokalemia requiring a close monitoring and an appropriate treatment.