Reactions of Acetylcholinesterase with Organophosphorus Insecticides
- 1. Department of Toxicology and Environmental Chemistry, The Islamic University-Gaza, Palestine
Abstract
The study characterized the reaction types of acetylcholinesterase (AChE) with different pesticides and suggested immediate medical treatments for poisoning cases. We retrospectively investigated the medical records of 120 poisoned cases. AChE activities were categorized into 3 groups. Extreme inhibition (EI), (307-769 u/L), severe inhibition (SI) (770-2888 u/L) Moderate inhibition (MI) (3000-4999 u/L), and less inhibition (LI) (5000-7000 u/L). These data were associated with occupational pesticides exposure to organophosphorus insecticides (OPI), carbamate insecticide (CI) s and general pesticides (GP). Medical treatments showed that class of EI did not show any cure signs with atropine administration whereas other classes (SI, MI and LI) showed positive responses with atropine administration. Class of EI showed positive sings with Toxogonin administration after several dose. These data suggested AChE in EI class has a strong affinity to bound with OP forming irreversible and/or stable complex, whereas in CI and GP, AChE has low affinity and form reversible non-stable complexes. It can be concluded that Toxogonin can be given to EI cases of AChE activity level as successful and immediate medical treatment for OP poisoning cases. On the other case atropine is the best antidote.
Keywords
• AChE activities
• Severe inhibition
• Irreversible reactio
Citation
El-Nahhal Y (2018) Reactions of Acetylcholinesterase with Organophosphorus Insecticides. J Pharmacol Clin Toxicol 6(2):1108.
IOINTRODUCTION
AChE is an important enzyme in the nervous system. It catalyzes the hydrolysis of the neurotransmitter acetylcholine (ACh) in the synaptic gaps. This enzyme is sensitive to react with many inhibitors such as organophosphorus insecticides and carbamate compounds. Its reactions with inhibitors may be associated with Ach accumulation in the synaptic gaps resulting in the appearance of cholinergic symptoms. This is in agreement with previous reports [1-5]. So far reactions of AChE with OPI have been reported [6], the author revealed several mechanisms. So far, AChE is sensitive to a wide number of pesticides and other chemical compounds and can be inhibited by them. The magnitude of inhibition is different from molecule to another [7-9]. The above mentioned studies were limited to toxicity or inhibition studies. Types of possible AChE reactions with OPs or pesticides in general was not investigated or remains in the primary stages. This investigation is concerned with the reaction types of AChE with organophosphorus and carbamate pesticides, with emphasis on electronegativity of atoms on the molecular structure
MATERIALS AND METHODS
Data Collection
About 120 medical reports of those medically visited the hospital for occupational pesticide treatments such as accidental poisoning, vomiting, diarrhea and headache during work were collected and analyzed. The medical file included results of acetylcholine esterase activity (AChE) measurement. Additionally, the medical files contained data of pesticides used during the medical treatment and the antidotes used for medical treatments.
Statistical Analysis
The collected data were analyzed using an Excel program. The pesticides used were arranged into different groups such as OPI, CI, and GP. AChE activity was subdivided into four groups as follows: group 1 (EI) [307-769 u/L], group 2 (SI) [770-2888 u/L, group 3 (MI) [3000-4982 u/L] group 4 (LI) [5000-7000 u/L] and normal range [7001-9776 u/L]. The pesticides found were listed in Groups.
RESULTS
Levels of AChE
Levels of AChE detected in blood serum of farmers occupationally exposed to different pesticides are shown in Table 1
Table 1: levels of AChE among farmers exposed to different pesticides during work.
| Type of inhibition | AChE levels | No cases | % | Pesticides found | LD50-range mg/kg |
| EI | 307-769 u/l | 15 | 12.82 | OP insecticides | 6-27 |
| SI | 770-2888 u/l | 16 | 13.68 | Carbamate insecticides | 8-30 |
| MI | 3000-4982 u/l | 10 | 8.55 | OP Pesticides | 1375 |
| LI | 5000-7000 u/l | 30 | 25.64 | All pesticides | >1500 |
| Normal range | 7001-9776 u/l | 46 | 39.31 | Control group | 00 |
| The name and chemical structure of pesticides found in EI and SI groups are shown in Table 2 | |||||
Proposed reactions of AChE with different pesticides
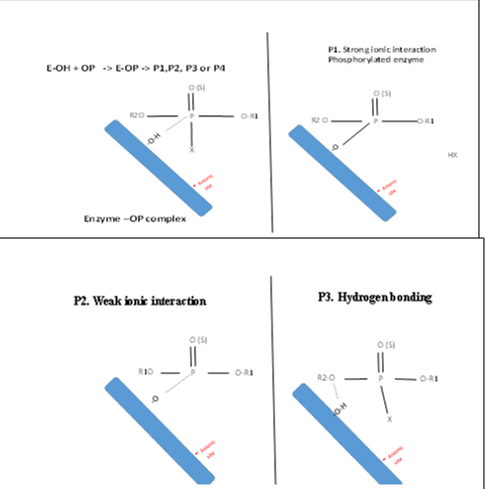
Pesticides in general can react with AChE in four different ways:
1. Strong ionic interaction between the anionic site on the AChE and the cationic Phosphonium ion in OP insecticides
2. Weak ionic interaction between the anionic site on the AChE and the cationic Phosphonium ion in OP
3. Hydrogen bonding between the hydrogen atoms of the AChE and O-atom in the OP
4. Hydrophobic/hydrophobic interaction between the hydrogen atoms in OP molecules and hydrogen atoms in esteratic site on the enzyme surfaces. Schematic diagram of these reactions are shown in Figure 1.
Figure 1 The possible reactions of AChE with Ops.
Medical treatments
The cases of AChE levels visited the main hospital in Gaza listed in Table 1 were treated with both atropine and Toxogonin, the EI cases and SI cases were treated with atropine but without significant progress. Then were give Toxogonin, few days later the level of AChE increased indication progress in the medical treatments. On the other hands MI and LI cases show tremendous medical progress from the first dose indication of weak interaction between pesticide and AChE.
DISCUSSION
The data presented in Table 1 shows the inhibition levels of AChE and the number of cases. The presented results clearly show extreme and severe inhibition levels among farmers. These levels are related to the high toxicity of applied pesticides found in the medical records of the farmers. These results are in accordance with recent levels [5,10]. Furthermore, the data in Table 2
|
Table 2: Name, chemical structures, electron withdrawing groups, and LD50. |
|||
|
Name |
Chemical structure |
(electron withdrawing groups) |
*LD50 mg/kg |
|
Azinophos-methyl |
|
3 |
12 |
|
Azinophos-ethyl |
3 |
9 |
|
|
fenamiphos |
|
4 |
6 |
|
Isofenphos |
3 |
20 |
|
|
methamidophos |
3 |
15.6 |
|
|
Cadusafos |
4 |
37.1 |
|
|
Glufosinate |
2 |
2000 |
|
|
Butamifos |
|
2 |
2000 |
|
Glyphosate |
1 |
5600 |
|
|
LD 50 values were collected from Tomlin 2000. |
|||
clearly show the chemical structure, number of electron with drawing atoms or groups and the LD50. It is obvious that the OP compounds containing aromatic and aliphatic groups. It can be noticed that LD50 value decreased (higher toxicity) as the OP compound contained electron withdrawing atoms or groups more that2. The highest toxicity was found with fenamiphos, which has four electron with drawing groups whereas the lowest toxicity was found with Glyphosate, which has the lowest number of electron with drawing groups, only one group.
These observation indicate the importance of electron withdrawing groups that attached to the phosphorus atom in the OP molecules. The explanation of these results is that electron with drawing group can create partial positive change on the phosphorus atom. As the number of electron with drawing groups increased in the OP molecules as in fenamiphos molecules, the potential formation of positive charge on the phosphorus atom increased leading to a possible formation of a phosphonium ion which can strongly react with AChE forming irreversible complexes as in the EI cases and SI (Table 1).
Furthermore, the degree of positive charges creation on the phosphorus atom, the degree of strength interaction between the OP molecules and AChE. These reactions are proposed in Figure 1.
It can be noticed that four reaction categories are proposed based on the degree of electronegativity of the attached groups with phosphorus. So far at highest electronegativity, the strongest reaction would be formed as in the cases of strong ionic interaction between the anionic site on the AChE and the positive charge in phosphorus atom. This type of reaction would occur in the following OPs (Azinophos-methyl, Azinophos-methyl, fenamiphos, Isofenphos). Furthermore, in the cases of lower electron withdrawing groups, weak ionic interaction may be formed (Figure 1). This may occur with the following Glufosinate Table 1. Moreover, at the lowest electron with drawing groups attached with the OP, a hydrogen bonding between the hydrogen atoms of the AChE and O-atom in the OP. The last option of reaction includes
Hydrophobic/hydrophobic interaction between the hydrogen atoms in OP molecules and hydrogen atoms in esteratic site on the enzyme surfaces these. This type of reaction may occur with many pesticides not only OP, carbamate but also others such as herbicides and fungicides with do not regularly attach AChE. These explanations agree with previous reports [6], who emphasized the influence of chemical structure, in the activity of OP. Additionaly, (Nair and Hunter 2004) [13] revealed that acetylcholinesterase has two active sites, an anionic site and an esteratic site. Moreover, Pohanka [1] constructed electrochemical biosensors using AChE, recently [11,12] developed AChE reactions for determination of OP residues in environmental samples and agricultural produces.
CONCLUSION
ChE reactions are important for the toxicity and ecology studies. It appeared that electron withdrawing groups attached with phosphorus atom are important to show the reaction either in toxicity or ecology studies. Four reaction types are found with AChE, these are listed above. EI cases are not positively responding to the medical care atropine whereas Toxogonin was effective for them. The other chemical reactions responded positively to atropine. It can be concluded that at extreme inhibited case atropine is not recommended Toxogonin is recommended.