Sudden-Onset Unilateral Sensorineural Hearing Loss in a Metastatic Lung Adenocarcinoma Patient Treated with Pembrolizumab: A Case Report and Review of the Literature
- 1. Department of Otolaryngology–Head and Neck Surgery, Saint Joseph University, Lebanon
- 2. Department of Otolaryngology–Head and Neck Surgery, Toulouse University Hospital, University Toulouse III- Paul-Sabatier, France
Abstract
Background: Immune checkpoint inhibitors, such as pembrolizumab, have significantly improved outcomes in advanced non-small cell lung carcinoma. While well-tolerated in most cases, sudden onset unilateral sensorineural hearing loss is a potential complication of checkpoint inhibitors rarely reported in the literature.
Case Presentation: We report the case of a patient in their 70s with a history of T3N0M0 lung adenocarcinoma treated with surgery and adjuvant chemotherapy, who experienced metastatic recurrence three years later and was placed on pembrolizumab-based systemic therapy. During maintenance monotherapy with pembrolizumab, the patient developed sudden onset right-sided sensorineural hearing loss. Systemic corticosteroid therapy led to significant improvement in the right-side hearing.
Conclusion: This case highlights a rare but potentially reversible presentation of sudden onset unilateral sensorineural hearing loss in the context of immune checkpoint inhibitor therapy. Early recognition and prompt intervention with systemic corticosteroids may help preserve hearing function. Clinicians should be aware of auditory symptoms in patients receiving checkpoint inhibitors, even during long-term maintenance.
Insights: Sudden-onset unilateral sensorineural hearing loss may represent a rare, immune-mediated adverse effect of pembrolizumab that can be reversible with prompt intervention.
Keywords
• Immune Checkpoint Inhibitors
• Adenocarcinoma
• Autoimmune
• Immunotherapy
• Lung Cancer
Citation
Makhoul M, Khneisser M, Ismail S (2025) Sudden-Onset Unilateral Sensorineural Hearing Loss in a Metastatic Lung Adenocarcinoma Patient Treated with Pembrolizumab: A Case Report and Review of the Literature. Ann Otolaryngol Rhinol 12(6): 1375.
ABBREVIATIONS
ICI: Immune Checkpoint Inhibitor; NSCLC: Non Small Cell Lung Carcinoma; PD-L1: Programmed Death Ligand 1; irAEs: Immune-Related Adverse Events;SNHL: Sensorineural Hearing Loss;PD-1: Anti-Programmed death 1; AIRED: Autoimmune Inner Ear Disease (AIED)
BACKGROUND
Immunotherapy with immune checkpoint inhibitors (ICIs) have become essential in the management of advanced non-small cell lung carcinoma (NSCLC), particularly in tumors expressing programmed death ligand 1 (PD-L1) [1]. ICIs can trigger inflammatory responses in any organ system, leading to toxicities collectively referred to as immune-related adverse events (irAEs). Although generally well tolerated, ICIs are most commonly associated with irAEs involving the endocrine, dermatologic, pulmonary, and gastrointestinal systems [2]. However, ototoxicity, particularly sudden-onset SNHL, has not been widely reported in association with ICIs therapy. To date, the literature contains very few documented cases of acute auditory dysfunction caused by ICIs, and the underlying pathophysiological mechanisms remain poorly understood. We present a rare case of sudden-onset unilateral SNHL occurring in association with the anti programmed death 1 (PD-1) antibody pembrolizumab in a patient with metastatic lung adenocarcinoma, that improved markedly following corticosteroid treatment.
CASE PRESENTATION
A patient in their 70s was referred to our tertiary care hospital after experiencing a sudden onset right-sided hearing loss. The medical history included hypertension, hypertriglyceridemia, and presbycusis. In 2019, he was diagnosed with an incidentally discovered right lower lobe pulmonary nodule during imaging for diverticulitis. Initial management consisted of observation and empirical antibiotics.
A positron emission tomography–computed tomography in August 2021 revealed isolated hypermetabolism of the lesion. Computed tomography guided biopsy confirmed a TTF1-positive non-small cell lung adenocarcinoma. PD-L1 expression was 5%, and no other mutations (EGFR, ALK, ROS1) were identified. Staging imaging revealed no distant metastases, and the patient underwent right lower lobectomy with lymph node dissection in December 2021. Pathology confirmed a 5.1 cm infiltrating adenocarcinoma, pT3N0R0. The patient received four cycles of adjuvant cisplatin and vinorelbine.
In 2023, surveillance imaging showed new ground glass nodules in the right upper and middle lobes. A diagnosis of metastatic recurrence was made. The patient received four cycles of carboplatin, pemetrexed, and pembrolizumab from July to September 2024. Due to asthenia, pemetrexed was discontinued after the fourth cycle, and pembrolizumab monotherapy was continued every six weeks from October 2024 onward.
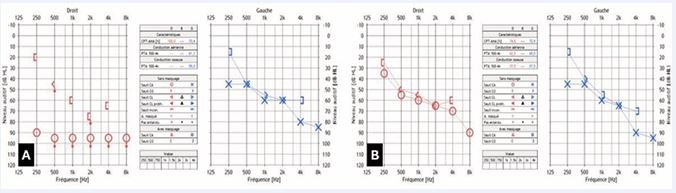
In June 2025, approximately 12 months after initiating pembrolizumab therapy and one week following the most recent cycle, the patient reported sudden onset of right sided hearing loss. There were no vestibular symptoms,ear pain, otorrhea, or any neurological, joint, or skin manifestations. Otoscopy was unremarkable. Audiometry revealed complete right hearing loss and moderate left sided SNHL [Figure 1A].
Figure 1 A: Audiogram showing complete right hearing loss and moderate left-sided sensorineural hearing loss B: Audiogram showing a significant improvement of the hearing on the right side with no change on the left side one week after initiating high dose oral prednisone.
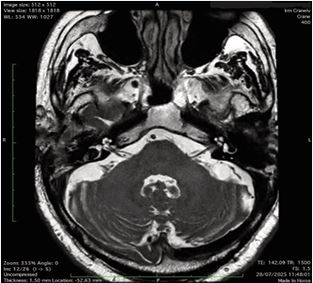
High-dose oral prednisone (1 mg/kg/day for 7 days) was initiated. At one-week follow-up, the patient reported marked improvement in right-sided hearing, confirmed by audiometry, while left-sided hearing remained unchanged [Figure 1B]. An MRI of the internal auditory canals was performed to rule out retro cochlear pathology, which was subsequently excluded [Figure 2]. In addition, an autoimmune and infectious workup was conducted and returned within normal limits.
Figure 2 Axial T2-weighted MRI of the inner ear showing no evidence of right retro cochlear pathology.
DISCUSSION
The present case of sudden-onset unilateral SNHL occurring after 12 months of pembrolizumab monotherapy highlights a rare but clinically significant immune-mediated ototoxicity in the setting of ICI therapy. In the literature, only few reports have described immune mediated ototoxicity associated with ICI therapy. Zibelman et al., described the first case of autoimmune inner ear disease (AIED) in a melanoma patient on pembrolizumab, which improved after intratympanic dexamethasone injections [3]. Additionally, Hobelmann and Fitzgerald, documented a case of severe bilateral SNHL after a single dose of pembrolizumab, with recovery following oral and intratympanic steroids [4].
A systematic review had been published in 2023 including 38 patients with ICI-induced hearing loss. In this review, the most cases occurred within a median of 3 months after ICI initiation, hearing loss was bilateral in approximately 69?% of patients, and improved in nearly 46?% after corticosteroid treatment [5]. This review also demonstrated that hearing loss frequently occurred in melanoma patients and was often associated with other irAEs [6]. Rosner et al., published a case series of melanoma patients treated with ICIs who developed cochleovestibular toxicity, including symptoms such as vertigo and tinnitus [7]. Furthermore, a case of unilateral AIED associated with nivolumab had been reported in a lung cancer patient, further highlighting the potential role of autoimmunity in the development of these events [8].
Unlike prior case reports, our patient’s unilateral presentation after long-term pembrolizumab therapy (12 months) is unique; most literature reports indicate earlier onset (within months). The close temporal association with the last infusion of pembrolizumab, significant audiometric improvement following systemic corticosteroids, absence of other causes on MRI, autoimmune and infectious workup, strongly suggest an ICI-induced autoimmune mechanism.
The pathophysiology of ICI-induced ototoxicity remains poorly understood. The main hypothesis suggests that PD-1 blockade triggers unregulated T-cell activation, leading to autoimmune inner ear inflammation, analogous to AIED [9]. This case reinforces that hypothesis and emphasizes that otologic irAEs may present well beyond initial treatment phases and remain potentially reversible when recognized early.
Our findings support the early use of high-dose systemic corticosteroids at symptom onset, as evidenced by hearing improvement within days, consistent with prior case report [10]. Given the uncommon nature of this irAE, we recommend that clinicians maintain a high index of suspicion for new auditory symptoms in patients receiving ICIs, regardless of treatment duration. Limitations of this case report include the absence of long-term follow-up audiometry beyond initial recovery. However, this case adds important information to a few reported cases, showing that ICI-induced SNHL can be reversible, and emphasizes the need for future studies to monitor this side effect.
CONCLUSION
This case contributes to the emerging evidence that ICIs can cause reversible immune-related ototoxicity. Clinicians should maintain a high index of suspicion for sudden hearing loss in patients on ICIs, even during prolonged maintenance therapy. Prompt audiologic evaluation and corticosteroid therapy may lead to functional recovery and avoid permanent disability.
DECLARATIONS
- Ethical approval: This study has obtained approval from the hospital ethical committee.
- Consent to Participate: Informed consent was obtained from the patient
- Consent for publication: Consent for publication was obtained from the patient
- Availability of data and materials: data are available from the corresponding author upon demand
- Competing interests: The authors declare that they have no conflict of interest
- Funding: This study was not supported by any funding
- Authors’ contributions: Mikhael Makhoul identified the case, collected clinical data, conducted the literature review, and drafted the initial manuscript. Myriam Khneisser contributed to the clinical evaluation, interpretation of findings, and provided substantial revisions to the manuscript. Salim Ismail supervised the study, verified the accuracy of the data, and offered critical intellectual input. All authors meet the ICMJE authorship criteria, have reviewed the manuscript, and approve its final version for submission.
REFERENCES
- Tang S, Qin C, Hu H, Liu T, He Y, Guo H, et al. Immune Checkpoint Inhibitors in Non-Small Cell Lung Cancer: Progress, Challenges, and Prospects. Cells. 2022; 11: 320.
- Haanen J, Obeid M, Spain L, Carbonnel F, Wang Y, et al. Management of toxicities from immunotherapy: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol. 2022; 33: 1217- 1238.
- Zibelman M, Pollak N, Olszanski AJ. Autoimmune inner ear disease in a melanoma patient treated with pembrolizumab. J Immunother Cancer. 2016; 4: 8.
- Kealan Hobelmann, Dennis Fitzgerald. A Case of PembrolizumabInduced Autoimmune Sensorineural Hearing Loss.
- Guven DC, Erul E, Kaygusuz Y, Akagunduz B, Kilickap S, De Luca R, et al. Immune checkpoint inhibitor-related hearing loss: a systematic review and analysis of individual patient data. Support Care Cancer. 2023; 31: 624.
- Rosner S, Agrawal Y, Sun DQ, Aygun N, Schollenberger MD, LipsonE, et al. Immune-mediated ototoxicity associated with immune checkpoint inhibitors in patients with melanoma. J ImmunotherCancer. 2020; 8: e001675.
- Lemasson J, Cuzzubbo S, Doucet L, Gounant V, Baroudjian B, Herms F, et al. Cochleovestibular toxicity induced by immune checkpoint inhibition: a case series. Eur J Cancer. 2019; 117: 116-118.
- Page JC, Gidley PW, Nader ME. Audiovestibular Toxicity Secondary to Immunotherapy: Case Series and Literature Review. J Immunother Precis Oncol. 2022; 5: 2-6.
- Ciorba A, Corazzi V, Bianchini C, Aimoni C, Pelucchi S, Skar?y?ski PH, et al. Autoimmune inner ear disease (AIED): A diagnostic challenge. Int J Immunopathol Pharmacol. 2018; 32: 2058738418808680.
- Stürmer SH, Lechner A, Berking C. Sudden Otovestibular Dysfunction in 3 Metastatic Melanoma Patients Treated With Immune Checkpoint Inhibitors. J Immunother. 2021; 44: 193-197.