Effects of Steviol Glycosides and Steviol on the Relative Abundance and Diversity of the Human Gut Microbiome in the Fecal Homogenates from Healthy Adults and Children
- 1. SP Advisors Inc, Chicago, USA
- 2. Department of Food Science, University of Massachusetts, USA
- 3. PureCircle Stevia Institute, PureCircle by Ingredion, USA
- 4. JNJ Corporation Services” should be changed to BRI Biopharmaceutical Research Inc.
Abstract
Steviol glycosides extracted from stevia rebaudiana plant are increasingly used as sweeteners. The minor steviol glycosides in the stevia extract, such as rebaudioside D and rebaudioside M, have superior sensory quality compared to the major components (e.g. rebaudioside A and stevioside). Due to the limited availability of rebaudiosides M and D in stevia leaf, they can be commercially produced by using enzymatic bioconversion and fermentation processes. The effect of steviol glycosides produced by different processes and their common metabolite steviol on human colonic bacteria is not adequately studied or published. There are limited research findings of the effect of artificial sweeteners, such as sucralose and Ace-K, on microbiomes. The key findings on artificial sweeteners are often extended, albeit incorrectly, to present the effect of all sweeteners including stevia, a natural sweetener on human microbiome. This study was designed to examine the potential influence of steviol glycosides and metabolite on the diversity and relative abundance of colonic bacteria genera in human fecal homogenates collected from adult male, female and young children.
During the in vitro biotransformation of steviol glycosides to steviol as a final common metabolite in human fecal homogenate, the metabolic fate of steviol glycosides and the effect of stevia metabolites on the bacterial population were investigated by employing the next-generation genomic sequencing and informatics to record the bacterial diversity and relative abundance in the fecal microbiota. From in vitro anaerobic incubation samples, 20 major bacteria genera were observed, accounting for a range of 75% to 85% of the freshly collected colonic populations in male, female and children. Based on hierarchical clustering analysis, beta diversity differences were noted in bacterial genera between adult male, female and children fecal homogenate incubation samples as anticipated to be influenced by dietary variables and age as previously recognized by other researchers. The relative abundance of colonic bacterial populations in the incubation samples were different from the corresponding raw stool samples due to the influence of enrichment medium during fecal homogenate metabolic incubation. However, the principal coordinate analysis of bacteria genus data indicated a general lack of differences in alpha and beta diversity between samples incubated for 72 hours in the presence and absence of steviol glycosides and steviol metabolite amongst fecal homogenates collected from adult male, female and children. The relative abundance data (%) of 30 bacteria genus showed some effect of experimental incubation time, but there was no apparent difference between the fecal homogenate samples incubated in the presence and absence of steviol glycosides and steviol. The adult male, female and children bacteria genera data from this study have contributed to a conclusion that steviol glycosides used at typical low concentrations as a non-nutritive sweetener did not result in observable alternations of the bacterial population.
Keywords
• Gut microbiome
• Steviol glycosides
• Fecal homogenates
• Sweetener
• Bacterial population
Citation
Purkayastha S, Kwok D (2026) Effects of Steviol Glycosides and Steviol on the Relative Abundance and Diversity of the Human Gut Microbiome in the Fecal Homogenates from Healthy Adults and Children. Ann Food Process Preserv 9(1): 1042.
INTRODUCTION
Steviol glycosides isolated from Stevia rebaudiana are a group of over 60 identified naturally occurring glycosides containing a common steviol aglycone [1,2]. Previous in vitro and ex vivo toxicology studies [3,4], have demonstrated that steviol glycosides are not absorbed from the gastrointestinal tract but undergo deglycosylation by bacterial hydrolysis to steviol as a final stable metabolite, which can either be excreted in stool or absorbed and metabolized to steviol glucuronide, which is excreted primarily via the urine in human. The rapid hydrolysis of steviol glycosides is the result of bacterial enzymatic activity in the lower bowel. The gut microbiome, which is amongst the most extensively studied of the human microbiomes, is highly diverse and contains thousands of different bacterial species [5,6]. The diverse community of bacteria is composed of a small number of abundant species plus many species found at low abundances [7]. One of the abundant bacterial family is the Bacteroidaceae, predominantly Bacteroides, that have previously been identified as being primarily responsible for the enteric biotransformation of steviol glycosides [4,8].
Steviol glycosides derived from stevia leaf extract have been increasingly used commercially as a natural low caloric sweetener along with artificial non-nutritive sweeteners in food and beverages. A review of published literature on the commonly used low caloric non-nutritive sweeteners including aspartame, saccharin, acesulfame K, and steviol glycosides and their influence on the diversity and abundance of the human gut microbiome revealed that limited amount of human data and the interpretation of animal experimental data did not adequately support a conclusive attribution between sweeteners and their influence on the human gut microbiome [9]. Another literature review [10], on the effects of sweeteners towards metabolic syndrome in the obese population characterized by insulin resistance and cardiovascular risks concluded that sweeteners could interact with the gut microbiota and intestinal sweet-taste receptors, thus considerations in daily intake, sweeteners composition and human metabolism are useful in considerations in their use in a sugar-free product.
Despite the historical safe use of steviol glycosides in small quantities commonly found in dietary exposure, detailed information on the influence of steviol glycosides and the common metabolite steviol on the beta- diversity and relative abundance of the human gut microbiome is limited. Hence, this study employed a well- established in vitro metabolic incubation protocol [11-13], involving the use of pooled human fecal homogenates to investigate a possible influence of steviol glycosides and the common final deglycosylation metabolite, steviol, on the gut microbiome diversity and relative abundance based on fecal materials collected from healthy adults and children donors. In this investigation, the use of in vitro pooled human fecal homogenates was designed to provide representative gut microbiome materials to control the complexity and interplay between individual variations in the gut microbiome due to diet and other confounding variables amongst the subjects.
The highly diverse human gut microbiome contains thousands of enteric bacterial species. Technological advances in high-throughput sequencing of genome have accelerated the analysis of the human gut microbiome since much of the enteric bacterial species cannot be readily cultured using standard laboratory culture techniques [14-16]. The most common sequencing approach to analyze the microbiome is amplicon analysis of the 16S ribosomal RNA (rRNA) gene, which was extensively used to compile most of the data collated by the Human Microbiome Project [17]. The annotation of the 16S method is based on the presumed association of the 16S rRNA gene with a taxa defined as an operational taxonomic unit (OTU), which are generally analyzed at the phyla or genera level with limited precision at the species level. The use of different sequencing methods to analyze the human fecal microbiome has been reviewed [16]. The authors concluded that whole genome shotgun sequencing (WGS) is more effective and accurate in defining a taxa at the species level. The WGS method uses sequencing with random primers to sequence overlapping regions of a genome. However, the WGS method generally requires sequencing a genome with high coverage which involves extensive data analysis and specialized database capability.
Within the children population, the gut bacterial composition changes readily during the first few years of life after birth, evolving with adaptation throughout the lifetime of the individual [18-21]. The gut microbiome is acquired at birth from the mother and is influenced by the method of delivery [18]. The development of the gut microbiota from birth to approximately 2.5 years of age showed that bacteria of the Firmicutes phylum generally dominated the early stages of the gut microbiota, whereas the introduction of solid foods promoted the establishment of a gut microbiome akin to adults, dominated by Firmicutes and Bacteroidetes in addition to low abundance of Proteobacteria and other enteric Gram negative bacteria [18,22,23]. In adults, the composition of the gut microbiome is dominated by members of the Bacteroidetes and Firmicutes, and to a lesser extent, Proteobacteria, Actinobacteria, Fusobacteria, Spirochaetes, Verrucomicrobia, and Lentisphaerae [21-27].
It has been described that by the time solid foods are introduced to infants, the gut microbiome has reached a significant level of diversity, including a high proportion of Bacteroidetes expressing metabolic enzymes including beta-glycosidases and beta-glucuronidases which are responsible for deglycosylation of steviol glycosides to steviol as a final metabolite [22,24,28]. As a result, the gut microbiome in children within a few months of birth would be expected to develop a capacity to hydrolyze steviol glycosides. Those steviol glycosides that are not completely hydrolyzed would simply transit through the intestines and excreted since steviol glycosides have been previously shown (3, 4, 7) to remain non-absorbed at normal consumption levels. Accordingly, in very young children with a lower capacity to metabolize steviol glycosides to steviol compared to adults, a lower systemic exposure of steviol would be expected relative to adults.
An increasing number of published articles have discussed the linkages of the human gut microbiome in a wide range of human diseases and conditions including inflammatory bowel disease [29], irritable bowel syndrome [30], obesity [31], type 2 diabetes [32], multiple sclerosis [33], Parkinson’s disease [34], autism [35], and colorectal cancer [36]. These studies revealed the association of a distinctive pattern in the diversity and relative abundance of the human gut microbiome in disease subjects compared with control healthy subjects and emphasized the need to investigate potential alterations in the gut microbiome in relations to the etiology of many human diseases.
MATERIALS AND METHODS
Healthy Adult and Children Human Fecal Donors
A summary of the experimental scheme is summarized in Figure 1. A total of six male adult donors aged between 22 to 58, six female adult donors aged between 23 to 60, and six children donors aged between 24 to 36 months were enrolled with reference to study inclusion and exclusion criteria established for this study.
Donors provided a self-declaration with absence of dietary disease/allergy, consumption of sweeteners, prebiotic/fiber and probiotic supplements within 2 weeks, recent history of gastrointestinal illness, history of Type 1 or Type 2 diabetes and cardiovascular disease, history of gastrointestinal or bariatric surgery, alcohol abuse, use of laxatives within 2 weeks, antibiotic therapy or chemotherapy within 3 months, inflammatory disorders, and not enrolled in weight-loss diet.
Fecal material collection and handling involved the use of a customized stool collection kit provided to each donor. Upon voiding, an estimated 10 g to 20 g wet weight of fecal material from each donor was immediately collected in a pre-labelled 100 mL screw-capped polypropylene specimen container pre-filled with 50 mL 0.01 M isotonic anaerobic phosphate buffer (pH 7.0) containing Brain Heart Infusion broth (BHI). The initial fecal- to-buffer ratio of the collected sample was determined based on the tare weight of the screw-capped fecal sample container with the initial volume of buffer used. Based on the initial dilution determined for each fecal material, a subsequent dilution with 0.01 M isotonic anaerobic phosphate buffer (pH 7.0) is performed to provide a 12.5x diluted fecal homogenate. All fecal materials were stored at room temperature before being used in homogenate preparation within 48 hours.
In Vitro Incubation of Steviol Glycosides in Pooled Human Fecal Homogenates
For this study, a commercial steviol glycoside mixture extracted from stevia leaf (A95, Lot No A95-27A) was provided by PureCircle™ for this study. The A95 stevia leaf extract was selected because it contained Reb M and Reb D steviol glycosides, which are extensively used in food and beverage applications for their superior sensory attributes. Metabolic incubation in the presence and absence of steviol glycosides (Lot A95) was performed over a period of 72 hours and post-incubation samples were stabilized for DNA metagenomic analysis to provide species diversity and relative abundance data.
Figure 1 Experimental scheme
Under anaerobic conditions, each of the 6 male, 6 female and 6 children fecal materials collected in 0.01 M isotonic phosphate buffer (pH 7.0) was homogenized by shaking and mixing with a disposable stirrer. Fecal materials from 2 subjects of the same gender or group were pooled (n =2 pooled) to provide 3 lots of pooled fecal homogenates of male adults, female adults, and children. So, total 9 fecal homogenates were used for incubation. The sample preparation of the pooled fecal homogenate had been previously published [11-13].
Steviol glycosides were ex vivo spiked and incubated at a concentration of 200 µg/mL in each of 3 lots of human fecal homogenate, each pooled from 2 subjects. Incubation was carried out over a time intervals of 0, 12, 24, 48 and 72 hours. Into each pre-labelled sample vial, 10 µL of a Test Article (A95) or the positive control (Reb A) or the negative control (no steviol glycosides) stock solution was accurately dispensed followed by the addition of the 0.5 mL pre-incubated fecal homogenate to each vial performed inside an anaerobic nitrogen hood purged with high purity nitrogen to ensure <1% oxygen level. Samples were capped, vortex mixed and incubated under an anaerobic environment at 37°C±5°C in a calibrated environmental chamber. Independent triplicate samples were prepared for each time point with each of three lots of pooled adult male, female and paediatric fecal homogenate. An aliquot of each incubation sample with or without Test article (A95) was stabilized for DNA metagenomic analysis to generate species diversity and relative abundance data.
Human Gut Microbiome Shotgun Metagenomic Sequencing
A proprietary BoosterShot™ assay (CoreBiome™, Minneapolis, USA) particularly suited for human fecal samples was used to provide shotgun metagenomic sequence data. BoosterShot™ is a proprietary shotgun sequencing approach based on converting random DNA sequences into direct gene and species-level markers to generate accurate taxonomic and functional profiles of the microbiome with species-level resolution. Pooled human fecal homogenates in the presence and absence of steviol glycosides at each of the incubation time points were transferred into a commercial OMNIgene™ GUT fecal collection tube to provide contamination-free storage and stabilization for ambient temperature storage for up to 8 weeks to allow ease of handling prior to frozen storage until BoosterShotTM NGS assay.
Statistical Analysis of relative abundance of microorganism
Statistical Analysis was conducted on bacterial abundance data of 30 microorganisms using General Linear Model (GLM) function of ANOVA menu in Minitab 19 software of Minitab LLC (https://www.minitab.com). The factors for the GLM analysis were three Groups (Male, Female, Paediatric), five incubation times (0, 12, 24, 48, and 72 h) and two treatments (with and without stevia). The abundance (in percentage) of microorganism (genus level) was used as response. Two-way interactions for each factor were included in the analysis and ANOVA result was used to eliminate insignificant factors. Model was retested until residuals showed normality and constant variance. Transformation of response was conducted when appropriate using Box-Cox transformation. A 95% confidence interval (p-value 0.05) was used to determine statistical significance. The analysis was performed for each microorganism separately.
RESULTS
Metabolism of Steviol Glycosides by Human Gut Microbiome
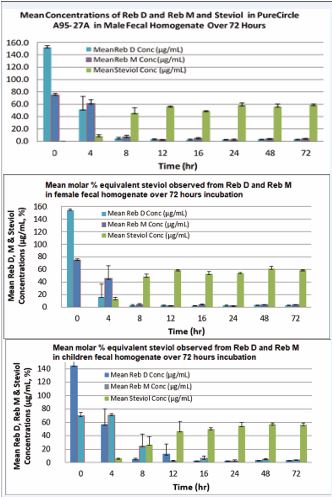
Metabolic incubation in the presence and absence of steviol glycosides was performed over a period of 72 hours and post-incubation samples were assayed for remaining steviol glycosides and steviol metabolite as previously described using a published LC-MS method [11-13]. Metabolic hydrolysis data had been reported elsewhere [2]. The mixture of steviol glycosides containing predominantly rebaudioside D and rebaudioside M were rapidly and nearly completely (>95%) deglycosylated during 12-16 hours of incubation in the pooled fecal homogenates collected from adult male, adult female and children. The formation of steviol as a final metabolite expressed as molar % equivalent over the incubation time intervals is summarized in Figure 2.
Figure 2 Pooled human fecal homogenate collected from adult male (top), adult female (middle) and children (bottom) donors showing rapid and near completely (>95%) deglycosylation of Rebaudioside D and Rebaudioside M and formation of molar equivalent % steviol metabolite observed during metabolic incubation over a 72-hour period.
Steviol Glycoside Influence on Human Gut Microbiome Diversity and Relative Abundance
From the metagenomic data on the pooled human fecal homogenate incubation samples, 20 major bacteria genera were observed and contributed to the overall relative abundance ranging from 75% to 85% of the gut microbiome observed in adult male, adult female and children.
Figure 3 Percent relative abundance of major human gut bacteria genera observed in pooled fecal homogenate from six males, six females, and six children incubated with and without steviol glycosides over a 72-hour period.
To investigate the potential influence of steviol glycosides and steviol on the beta diversity of the human gut microbiome, the % relative abundance of the most abundant bacterial genera observed in adult male, adult female and children pooled fecal homogenates over the metabolic incubation period are presented in Figure 3. Differences in beta diversity in the gut microbiome between adult male, adult female and children were observed in this study, which is likely an influence of dietary [26], and age [18], differences between the study groups as previously discussed in the literature. In this study, no marked differences in the alpha and beta diversity of the human gut microbiome were observed when comparing the presence and absence of steviol glycosides within adult male, adult female and children fecal microbiome samples over a metabolic incubation period of 72 hours (Figure 4).
Figure 4 No apparent differences in the alpha and beta diversities observed in the pooled fecal homogenate microbiomes in male, female and children materials during metabolic incubation in the presence and absence of steviol glycosides over a 72-hour period.
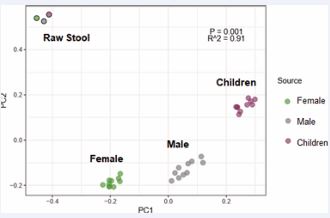
Figure 5 Principal coordinate analysis (PCoA) of bacterial composition between male, female and children pooled fecal homogenates
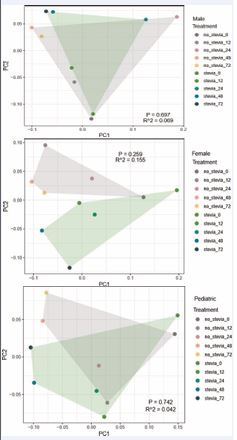
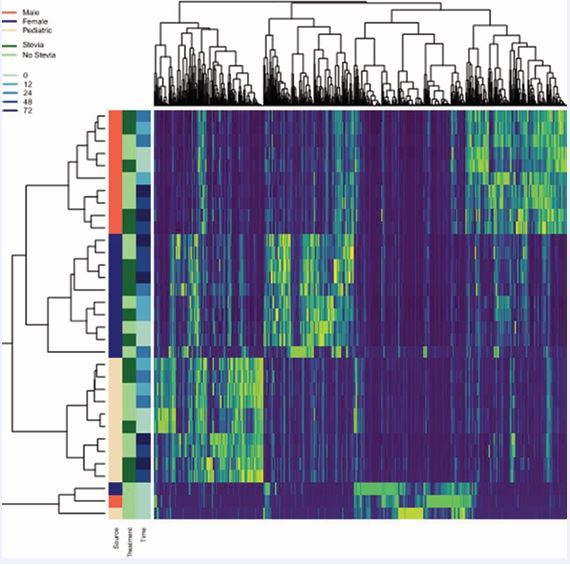
Principal coordinate analysis (PCoA) of the bacterial compositions observed in the raw stool samples and the corresponding pooled fecal homogenate samples of adult males, adult females and children is presented in Figure 5. The differences observed amongst different groups are likely due to the differences in diet and age. The raw stool materials without dilution by incubation medium were observed to be qualitatively different from the incubation samples at a 1:50 dilution in the BHI broth, which likely have resulted from enrichment during the metabolic incubation period. Nevertheless, additional hierarchical clustering analysis based on PCoA plots of the incubation samples in the presence and absence of steviol glycosides indicated the lack of steviol glycoside influence on the pooled fecal homogenate microbiome composition in adult male, adult female and children (Figure 6 and Figure 7), as indicated by their relative high p-values, low R-squared values and the overlapping coordinates between treatments in the presence and absence of steviol glycosides.
Figure 6 Principal coordinate analysis (PCoA) of bacterial composition in male (top), female (middle) and children (bottom) pooled fecal homogenates in the presence and absence of steviol glycosides over the 72-hour incubation time intervals.
To evaluate whether steviol glycosides influenced the bacteria genera relative abundance data in a statistically significant manner, ANOVA data from each of the bacterial genera relative abundance revealed significant differences at 95% confidence interval (P-value <0.05) over the incubation times and between adult male, adult female and children study groups. These differences observed across the bacteria genera agrees with the expected changes in bacteria relative abundances during metabolic incubation, and between pooled adult male, adult female and children fecal homogenates. However, differences in the relative abundance of each bacteria genus were generally not significantly different between the presence and absence of steviol glycosides indicating that steviol glycosides at the relatively low concentration evaluated in this study to reflect a low dietary consumption level did not impact on bacteria relative abundance. ANOVA data is in agreement with PCoA analysis supporting a conclusion that steviol glycosides did not influence the bacteria genera relative abundance.
Figure 7 Hierarchical clustering analysis based on PCoA plots of the incubation samples in the presence and absence of steviol glycosides indicated the lack of steviol glycoside influence on the pooled fecal homogenate microbiome composition in adult male, adult female and children.
DISCUSSION
A study on the short-term and long-term macronutrients influences on the relative abundance of the human gut microbiome have been published [37]. For instance, short-term consumption of a predominant animal-based diet increased the relative abundance of bile-tolerant Alistipes, Bilophila, and Bacteroides, while decreased Firmicutes such as Roseburia, Eubacterium rectale, and Ruminococcus bromii associated with metabolism of plant polysaccharides. The study revealed that microbes transiently colonize the human gut microbiome and can rapidly respond to changes in dietary components. The significance of diet in shaping the human gut microbiome has been reviewed [38]. There is a consensus that dietary alterations can induce large, temporary and predictable shifts in microbial populations within 24 hours and the authors suggested that dietary approach can be utilized to manipulate bacteria genera populations responsible for host immune response and metabolic parameters with broad potential applications for human health.
Specifically, dietary intake of low-caloric non-nutritive sweeteners and their potential impact on the gut microbiome have been reviewed and studied in preclinical models [39]. The authors concluded that the use of sweeteners in animal models have provided evidence of putative unfavorable effects on the gut microbiome and metabolic endpoints. In contrast, a recent review of published data from double blind randomized controlled clinical trials involving non nutritive sweeteners, including aspartame, acesulfame potassium, sucralose, saccharin and stevia did not reveal a meaningful attribution of non-nutritive sweeteners with alternations on the relative abundance and diversity of the human gut microbiome and metabolic health endpoints [40-44]. Furthermore, asides from the evaluation of relative distribution and diversity of the gut microbiome, the concept of “Keystone Species” of gut bacteria which can exert key influences responsible for individuality of the gut microbiome and health status have been proposed [45]. For instance, Bacteroides fragilis and Bacteroides stercosis have a disproportionate influence on the structure of the gut microbiome even though these species are only found in moderate abundance. Importantly, the presence of both species is associated with an increased risk of colon cancer [46]. These studies have highlighted the complex multivariate roles of the human gut microbiome in human health. The interplay between dietary influences and the bacteria keystone species associated with the status of a specific human health condition must be taken into consideration during interpretation of clinical data when evaluating the influence of steviol glycosides on the gut microbiome and its potential influence on human health.
In vitro human fecal homogenate culture conditions have also been previously evaluated to determine the impact of culture media conditions on microbial growth [47]. The potential health effects of four bacterial culture media including the use of a gut microbiota medium, a bacterial growth medium, a brain-heart infusion broth, and a fastidious anaerobic broth were investigated based on the formation of short-chain fatty acids under each in vitro culture conditions. The results from this study indicated that all four in vitro culture media exhibited different effects on the metabolism and composition of the human fecal microbiota. The limited number of published studies on human fecal homogenate culture conditions provided an initial understanding that the relative abundance and diversity of the fecal microbiome are susceptible to alterations by in vitro culture media conditions, hence, relevant culture conditions and appropriate study designs with experimental controls are required to allow valid interpretation of results. Clearly, more research is warranted.
An in vitro and in vivo study on the effects of synthetic sweeteners acesulfame potassium (AceK), saccharin, and sucralose, including the sweetener stevia, have also been reported [48]. The authors concluded that the synthetic and non-nutritive sweeteners all exerted strong bacteriostatic effects on the gut bacterial phyla Bacteroidetes, Firmicutes and Proteobacteria (E. coli). For sucralose, the authors reported an IC50 at 58.4 mM, equivalent to 23 mg/ mL, which is 100 to 1000-fold of the levels found in commercial soda drink products labeled from 19 to 380 ug/mL. Similarly, the authors studied saccharin, AceK, and Reb A at 2.5% (25,000 ug/mL), which is in far excess of the typical 100 ug/mL concentrations of these sweeteners labelled in commercial soda drink products. Therefore, it is necessary to interpret the results from this study with caution with respect to the excessively high sweeteners concentrations studied and the relevant use of mouse gut microbiome data for inference to human gut microbiome.
In the current in vitro metabolic incubation investigation conducted under relevant experimental parameters, the results clearly showed that both Reb D and Reb M along with other minor glycosides in a stevia leaf extract were rapidly deglycosylated by the gut microbiome to steviol as a final major metabolite within 12 hours in a comparable manner in healthy adults (male and female) and in young children.
Beta diversity data from this study indicated that Bifidobacterium spp. is a predominant bacteria found in the fecal homogenate incubation samples from adult male, adult female and children. In addition, Providencia was also a predominant bacterium in both adult male and female, while Enterococcus spp. was observed in the children between 2 to 3 years of age. Amongst these predominant bacteria genera in the fecal homogenate incubation samples, Bifidobacterium spp. has been recognized as an important human gut microbiome genus in mediating beta glucosidase activity [49], and the common predominant presence of Bifidobacterium spp in adult male, adult female and children fecal samples from the current investigation is in agreement with the observed functional efficiency of the human gut microbiome in the metabolic clearance of steviol glycosides.
CONCLUSION
The effects of steviol glycosides and their final common metabolite, steviol, on the human fecal homogenates indicated that the 20 most abundant bacteria genera were commonly observed in the pooled fecal homogenates collected from male, female and children. The observed abundant bacteria genera contributed to the overall abundance ranging from 75% to 85% of the diverse colonic population found in the representative fecal homogenate preparation. During the 72-hour metabolic incubation period, the overall % microbial abundance in the study preparations was observed to remain comparable between male, female and children. In addition, the potential influence of steviol glycosides and steviol on beta diversity of the microbial population was further evaluated and the % relative abundance of 22 major bacteria genera observed between male, female and children samples remained comparable over the entire metabolic incubation period.
Beta diversity data from this study indicated that Bifidobacterium spp. is a common predominant species found in representative fecal homogenates from male, female and children. Amongst the predominant bacteria genera, Bifidobacterium spp. has been recognized as an important human gut microbiome genus in mediating beta-glucosidase activity, and the common predominant presence of Bifidobacterium spp in male, female and children fecal samples are in the agreement with the observed functional efficiency of the human gut microbiome in deglycosylation of steviol glycosides. ANOVA (P-value <0.05) data from each of the most abundant 30 bacterial genera revealed non-significant differences in the relative abundance between steviol glycoside and placebo control, indicating that steviol glycosides at the relevantly low concentration evaluated in this study to reflect a low dietary consumption level did not reveal an observable impact on bacteria relative abundance. Hierarchical clustering analysis and PCoA plots of the incubation samples in the presence and absence of steviol glycosides and steviol indicated the lack of influence on the gut microbiome composition amongst the representative male, female and children pooled fecal homogenates.
CONFLICT OF INTEREST STATEMENT
The authors declare that no known competing financial interests or personal relationships that could have influenced the work and results reported in this paper. BRI Biopharmaceutical Research Inc. (a Frontage Laboratory company) is a pharmaceutical research laboratory commissioned to conduct this study for Purecircle™.
FUNDING STATEMENT
This work was supported by research and development funding from PureCircle USA Inc (PureCircle by Ingredion, Illinois, USA).
ACKNOWLEDGEMENT
The authors would like to thank Mr. Chandler Zhang and other BRI Pharma (Vancouver, Canada) researchers for their assistance in the metabolic incubation study and Divergen (Previously Corebiome, St. John, Minnesota) scientists for metagenomic analysis. Special thanks to Patrick Tendick and William Anthony of Ingredion (Bridgewater, New Jersey) for their help with the statistical analysis.
REFERENCES
- Joint Food and Agricultural Organization (FAO) - World Health Organization (WHO) 91st Joint Expert Committee on Food Additives, Rome. 2021; 26: 1-48.
- Purkayastha S, Kwok D. Metabolic fate in adult and pediatric population of steviol glycosides produced from stevia leaf extract by different production technologies. Regul Toxicol Pharmacol. 2020; 116: 104727.
- Koyama E, Kitazawa K, Ohori Y, Izawa O, Kakegawa K, Fujino A, et al. In vitro metabolism of the glycosidic sweeteners, stevia mixture and enzymatically modified stevia in human intestinal microflora. Food Chem Toxicol. 2003; 41: 359-374.
- Renwick AG, Tarka SM. Microbial hydrolysis of steviol glycosides.Food Chem Toxicol. 2008; 46: S70-S74.
- Yatsunenko T, Rey FE, Manary MJ, Trehan I, Dominguez-Bello MG, Contreras M, et al. Human gut microbiome viewed across age and geography. Nature. 2012; 486: 222-227.
- Gill SR, Pop M, Deboy RT, Eckburg PB, Turnbaugh PJ, Samuel BS, et al. Metagenomic analysis of the human distal gut microbiome. Science. 2006; 312: 1355-1359.
- Lynch MD, Neufeld JD. Ecology and exploration of the rare biosphere. Nat Rev Microbiol. 2015; 13: 217-229.
- Gardana C, Simonetti P, Canzi E, Zanchi R, Pietta P. Metabolism of stevioside and rebaudioside A from Stevia rebaudiana extracts by human microflora. J Agric Food Chem. 2003; 51: 6618-6622.
- Lobach AR, Roberts A, Rowland IR. Assessing the in vivo data on low/ no-calorie sweeteners and the gut microbiota. Food Chem Toxicol. 2019; 124: 385-399.
- Gómez-Fernández AR, Santacruz A, Jacobo-Velázquez DA. The complex relationship between metabolic syndrome and sweeteners. J Food Sci. 2021; 86: 1511-1531.
- Purkayastha S, Pugh G Jr, Lynch B, Roberts A, Kwok D, Tarka SM Jr. In vitro metabolism of rebaudioside B, D, and M under anaerobic conditions: comparison with rebaudioside A. Regul Toxicol Pharmacol. 2014; 68: 259-268.
- Purkayastha S, Bhusari S, Pugh G Jr, Teng X, Kwok D, Tarka SM. In vitro metabolism of rebaudioside E under anaerobic conditions: Comparison with rebaudioside A. Regul Toxicol Pharmacol. 2015; 72: 646-657.
- Purkayastha S, Markosyan A, Prakash I, Bhusari S, Pugh G Jr, Lynch B, et al. Steviol glycosides in purified stevia leaf extract sharing the same metabolic fate. Regul Toxicol Pharmacol. 2016; 77: 125-133.
- Kuczynski J, Lauber CL, Walters WA, Parfrey LW, Clemente JC, Gevers D, et al. Experimental and analytical tools for studying the human microbiome. Nat Rev Genet. 2011; 13: 47-58.
- Stewart EJ. Growing unculturable bacteria. J Bacteriol. 2012; 194: 4151-4160.
- Ranjan R, Rani A, Metwally A, McGee HS, Perkins DL. Analysis of the microbiome: Advantages of whole genome shotgun versus 16S amplicon sequencing. Biochem Biophys Res Commun. 2016; 469: 967-977.
- Human Microbiome Project Consortium. Structure, function and diversity of the healthy human microbiome. Nature. 2012; 486: 207- 214.
- O’Toole PW, Claesson MJ. Gut microbiota: changes throughout the lifespan from infancy to elderly. Int Dairy J. 2010; 20: 281-291.
- Koenig JE, Spor A, Scalfone N, Fricker AD, Stombaugh J, Knight R, et al. Succession of microbial consortia in the developing infant gut microbiome. Proc Natl Acad Sci U S A. 2011; 108: 4578-4585.
- Institute of Medicine (US) Food Forum. The Human Microbiome, Diet, and Health: Workshop Summary. Washington (DC): National Academies Press (US). 2013.
- Kostic AD, Howitt MR, Garrett WS. Exploring host-microbiota interactions in animal models and humans. Genes Dev. 2013; 27: 701-718.
- Spor A, Koren O, Ley R. Unravelling the effects of the environment and host genotype on the gut microbiome. Nat Rev Microbiol. 2011; 9: 279-290.
- Lloyd-Price J, Abu-Ali G, Huttenhower C. The healthy human microbiome. Genome Med. 2016; 8: 51.
- Palmer C, Bik EM, DiGiulio DB, Relman DA, Brown PO. Developmentof the human infant intestinal microbiota. PLoS Biol. 2007; 5: e177.
- Ley RE, Hamady M, Lozupone C, Turnbaugh PJ, Ramey RR, Bircher JS, et al. Evolution of mammals and their gut microbes. Science. 2008; 320: 1647-1651.
- Power SE, O’Toole PW, Stanton C, Ross RP, Fitzgerald GF. Intestinalmicrobiota, diet and health. Br J Nutr. 2014; 111: 387-402.
- Borewicz K, Smidt H. Ecology of the human microbiome. In TheHuman Microbiome Handbook. DEStech Publications. 2016: 9-34.
- Dabek M, McCrae SI, Stevens VJ, Duncan SH, Louis P. Distribution of beta-glucosidase and beta-glucuronidase activity and of beta- glucuronidase gene gus in human colonic bacteria. FEMS Microbiol Ecol. 2008; 66: 487-495.
- Bull MJ, Plummer NT. Part 1: The Human Gut Microbiome in Health and Disease. Integr Med (Encinitas). 2014; 13: 17-22.
- Kennedy PJ, Cryan JF, Dinan TG, Clarke G. Irritable bowel syndrome: a microbiome-gut-brain axis disorder? World J Gastroenterol. 2014; 20: 14105-14125.
- Baothman OA, Zamzami MA, Taher I, Abubaker J, Abu-Farha M. The role of Gut Microbiota in the development of obesity and Diabetes. Lipids Health Dis. 2016; 15: 108.
- Karlsson FH, Tremaroli V, Nookaew I, Bergström G, Behre CJ, Fagerberg B, et al. Gut metagenome in European women with normal, impaired and diabetic glucose control. Nature. 2013; 498: 99-103.
- Jangi S, Gandhi R, Cox LM, Li N, von Glehn F, Yan R, et al. Alterations of the human gut microbiome in multiple sclerosis. Nat Commun, 2016: 1-11.
- Miraglia F, Colla E. Microbiome, Parkinson’s Disease and MolecularMimicry. Cells. 2019; 8: 222.
- Kang DW, Adams JB, Coleman DM, Pollard EL, Maldonado J, McDonough-Means S, et al. Long-term benefit of Microbiota Transfer Therapy on autism symptoms and gut microbiota. Nature. 2019; 9: 2581-2590.
- Ahn J, Sinha R, Pei Z, Dominianni C, Wu J, Shi J, et al. Human gut microbiome and risk for colorectal cancer. J Natl Cancer Inst. 2013; 105: 1907-1911.
- David LA, Maurice CF, Carmody RN, Gootenberg DB, Button JE,Wolfe BE, et al. Diet rapidly and reproducibly alters the human gutmicrobiome. Nature. 2014; 505: 559-563.
- Singh RK, Chang HW, Yan D, Lee KM, Ucmak D, Wong K, et al. Influence of diet on the gut microbiome and implications for human health. J Transl Med. 2017; 15: 73.
- Garcia K, Ferreira G, Reis F, Viana S. Impact of Dietary Sugars on Gut Microbiota and Metabolic Health. Diabetology. 2022; 3: 549-560.
- Sievenpiper JL, Purkayastha S, Grotz VL, Mora M, Zhou J, Hennings K, et al. Dietary Guidance, Sensory, Health and Safety Considerations When Choosing Low and No-Calorie Sweeteners. Nutrients. 2025; 17: 793.
- Kwok D, Scott C, Strom N, Au-Yeung F, Lam C, Chakrabarti A, et al. Comparison of a Daily Steviol Glycoside Beverage compared with a Sucrose Beverage for Four Weeks on Gut Microbiome in Healthy Adults. J Nutr. 2024; 154: 1298-1308.
- Gauthier E, Milagro FI, Navas-Carretero S. Effect of low-and non- calorie sweeteners on the gut microbiota: A review of clinical trials and cross-sectional studies. Nutrition. 2024; 117: 112237.
- Ruiz-Ojeda FJ, Plaza-Díaz J, Sáez-Lara MJ, Gil A. Effects of Sweeteners on the Gut Microbiota: A Review of Experimental Studies and Clinical Trials. Adv Nutr. 2019; 10: S31-S48.
- Singh G, McBain AJ, McLaughlin JT, Stamataki NS. Consumption of the Non-Nutritive Sweetener Stevia for 12 Weeks Does Not Alter the Composition of the Human Gut Microbiota. Nutrients. 2024; 16: 296.
- Fisher CK, Mehta P. Identifying keystone species in the human gut microbiome from metagenomic timeseries using sparse linear regression. PLoS One. 2014; 9: e102451.
- Moore WE, Moore LH. Intestinal floras of populations that have a high risk of colon cancer. Appl Environ Microbiol. 1995; 61: 3202-3207.
- Yousi F, Kainan C, Junnan Z, Chuanxing X, Lina F, Bangzhou Z, et al. Evaluation of the effects of four media on human intestinal microbiota culture in vitro. AMB Express, 2019; 9: 69.
- Wang QP, Browman D, Herzog H, Neely GG. Non-nutritive sweeteners possess a bacteriostatic effect and alter gut microbiota in mice. PLoS One. 2018; 13: e0199080.