Hernia Mesh is Contraindicated in Patients with Peritoneal Metastases
- 0. Program in Peritoneal Surface Oncology, MedStar Washington Cancer Institute and MedStar Washington Hospital Center, USA
ABSTRACT
Background: Mesh has been used to reinforce a fascia repair in hernia surgery with good results. The mesh becomes infiltrated by fibrous tissue to add strength to the repair.
Methods: Recently, surgery to prevent peritoneal metastases has emerged as an important new treatment modality in gastrointestinal cancer patients. Patients with peritoneal metastases who have mesh repair of a hernia are in danger of cancers cells becoming entrapped and progressing in the hernia mesh.
Results: Two patients had undergone hernia repair with mesh and subsequently required cytoreductive surgery for the treatment of peritoneal metastases. The mesh greatly complicated the surgery by making it more traumatic. This was due to the infiltration of scar tissue, but also due to the infiltration of cancer cells into the mesh.
Conclusions: In a hernia repair with mesh, surgery for peritoneal metastases becomes more complex and has increased morbidity. Hernia mesh should not be used in patients who subsequently require cytoreductive surgery.
KEYWORDS
Tumor cell entrapment; Carcinomatosis; Pseudomyxoma peritonei; Malignant peritoneal mesothelioma; Rectal cancer.
CITATION
Sugarbaker PH (2018) Hernia Mesh is Contraindicated in Patients with Peritoneal Metastases. JSM Surg Proced 1(1): 1001
INTRODUCTION
In the past, patients with peritoneal metastases from gastrointestinal cancer or malignant peritoneal mesothelioma were categorized as terminal. Palliative chemotherapy and best supportive care was the suggested treatment. In the last 20 years, data has become available showing that peritoneal metastases can be cured. The treatments that show greatest benefits are cytoreductive surgery combined with hyperthermic perioperative chemotherapy (HIPEC). As a result of these successful treatments in selected patients a large number of surgical procedures in patients with peritoneal metastases are now being performed. A proportion of these patients have had mesh placed with peritoneal metastases present most frequently when the first manifestation of the peritoneal metastases was a new onset hernia [1]. In this manuscript, we report our experience with mesh placement in two patients who had peritoneal metastases and went on at a later time to require definitive cytoreductive surgery and HIPEC. The potential for peritoneal metastases lodged within the intensely fibrotic mesh and the requirement for complete removal of the mesh created postoperative problems. From our experience with these two patients combined with a review of the literature, we conclude that hernia mesh is contraindicated in patients with peritoneal metastases, patients with positive cytology for cancer, or patients at high risk for the development of peritoneal metastases.
CASE PRESENTATION
Patient 1
This 83 year old man complained of a left inguinal hernia. A left inguinal hernia repair was performed laparoscopically with the placement of mesh in June of 2016. Shortly thereafter, the patient began complaining of rectal bleeding. In July of 2016, he had a colonoscopy which revealed a rectosigmoid colon cancer. A surgical procedure to remove this cancer was performed in July of 2016 but the resection was compromised because of a diffuse fibrotic reaction within the pelvis, possibly due to an extensive cancer but also related to the prior placement of hernia mesh. The resection was incomplete with an R1 margin on the distal rectum. Postoperatively, the patient developed deep vein thrombosis with a pulmonary embolus. He required hospitalization for 4 weeks because of pelvic sepsis.
Follow-up CT performed in July of 2017 showed a normal liver, a large paraostomy hernia, and a diffuse mass emanating from the mid-rectum that extended to the left pelvic sidewall. The patient was symptomatic from bloody mucus continuously leaking from his residual rectum.
It was decided that a repeat attempt at resection of his rectal cancer should occur. On July 27, 2017, the patient had exploratory laparotomy, complete resection of the mesh withinthe left inguinal hernia region with a left orchiectomy. Access to the pelvis was extremely difficult. The distal portion of the left ureter was resected and the pelvic peritoneum with multiple cancer nodules resected. Extensive fibrosis within the left side of the pelvis, in and around the hernia mesh greatly increased the complexity of the surgery. Cancer was removed from the rectal stump. Residual disease was left deep in the pelvis.
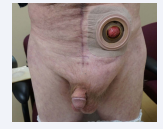
Postoperatively, the patient recovered well with some delay in the return of normal urinary tract function. He received definitive radiotherapy to the residual disease deep in his pelvis. This was facilitated by a rectus abdominis muscle flap used to separate small bowel from the rectal mass. He developed a large and uncomfortable hernia at the site of mesh removal that will require repair at some time in the future (Figure 1).
Figure 1 Large left inguinal hernia that resulted from removal of a mesh placed at a prior repair.
Patient 2
This 40 year old woman had symptoms of a left inguinal hernia in June of 2017. There was a laparoscopic repair with mesh. At the time of the laparoscopy, nodules were seen in the pelvis and these returned as malignant peritoneal mesothelioma. On September 21, 2017, the patient underwent a 9-hour cytoreductive surgical procedure that included a greater omentectomy, cholecystectomy, appendectomy, pelvic peritonectomy, and hysterectomy with bilateral salpingo-oophorectomy. During the dissection of the left pelvis there was a massive scar surrounding the hernia mesh within the left inguinal and left pelvic region. In removing the mesh there was an arteriotomy made in the left external iliac artery. The vein was dissected free without damage. The external iliac artery was repaired without incident. The mesh was completely extracted. The patient did receive hyperthermic intraperitoneal chemotherapy with cisplatin and doxorubicin and ifosfamide intravenously.
Postoperatively, it was noted by venous Doppler studies that the external iliac artery had thrombosed and blood supply to the leg was through collateral vessels below the inguinal ligament. Pathology report showed epithelial-type malignant peritoneal mesothelioma infiltrating the pelvic peritoneum and the mesh remaining from the prior left inguinal hernia repair.
Patient has some chronic pain in her left groin region. The hernia has not recurred and she is currently free of disease.
In patient 1, surgery to remove an upper rectal cancer was compromised as a result of the fibrosis surrounding the mesh in the left inguinal region. This was a large piece of mesh measuring approximately 12 cm x 9 cm. A second surgery to remove progressive disease at the top of a Hartmann closure of the rectum was also severely compromised by the chronic fibrosis in and around the left ureter, left pelvic sidewall and left common and external iliac artery and vein. At this second operation complete removal of the mesh resulted in a large and symptomatic hernia.
In patient 2, the mesh was infiltrated by malignant peritoneal mesothelioma. Complete removal of the mesh resulted in damage to the external iliac artery which later went on to thrombose. Fortunately, arterial reconstruction was not necessary because of extensive collaterals that developed.
DISCUSSION
Mesh makes subsequent surgery more difficult and more dangerous
There can be no doubt that an abdominal wall mesh makes subsequent surgical interventions more dangerous. In both patients, there were adverse events associated with surgery that was required in the area of prior hernia mesh placement. Patel and coworkers reported bowel resection or enterotomy in 5 of 18 (27%) patients who had a bowel obstruction after laparoscopic ventral hernia repair with mesh [2]. Sharma reported that previously implanted intraperitoneal mesh increases the morbidity during a re-laparoscopy [3,4]. This was associated with extensive adhesion formation caused by the intraperitoneal placement of mesh.
Mesh can complicate subsequent decisions in oncologic management
The presence of mesh in a cancer patient does complicate management. Audollent and colleagues reported that patients with intraabdominal mesh may have a falsely positive 18F-FDGPET/CT. The presence of the mesh may result in a false-positive radiologic finding in some patients with peritoneal metastases. Not only the PET scan but also CT or MRI cannot distinguish cancer regrowth from a heavily fibrosed hernia mesh. Extensive resections of mesh that does not contain malignancy in cancer patients with the surgeon wanting to remove all of the recurrent disease is completely possible. Also, oncologic problems with mesh are caused by the incorporation of cancer cells within the mesh. Patient 2 had extensive infiltration of the mesh by malignant peritoneal mesothelioma necessitating complete removal of this foreign body. Schwartzberg reported recurrence of a colonic adenocarcinoma in the mesh used for ventral hernia repair [5]. If mesh is present in a patient having definitive surgery for peritoneal metastases, the mesh within the peritoneal cavity or within a closure of the abdominal wall should be completely removed. Unfortunately, a problem that occurs intraoperatively with mesh present is the difficulty of distinguishing fibrous in growth into the mesh from cancerous involvement of the mesh. Intraoperative frozen section biopsies are usually not helpful. Probably the best recommendation is that mesh inserted whenfree intraperitoneal cancer cells (peritoneal metastases) were present must always be removed completely at any reoperative procedure. More discrimination, in terms of mesh removal, is possible if the mesh was placed at a distant point in time before the peritoneal metastases were present.
Mesh from an inguinal hernia repair causes increased surgical trauma
Sugarbaker studied cytoreductive surgery in patients who had prior placement of mesh in an inguinal hernia defect. The inguinal hernia had led to a diagnosis of mucinous appendiceal peritoneal metastases at the time of a prior inguinal hernia repair. If at the prior inguinal hernia repair no mesh was used at the time of definitive cytoreductive surgery, there was no damage to vas deferens, testicular artery, or inferior deep epigastric artery and vein in 5 patients. However, if mesh had been used at the time of prior inguinal hernia repair, 4 of 5 patients had resection of the vas deferens, 5 of 5 patients had resection of the testicular artery, and 3 of 5 patients required resection of the inferior deep epigastric artery and vein. These data strongly suggest that surgery in the inguinal region is more dangerous in patients who have had prior mesh placement at a prior inguinal hernia repair [6]. As seen in patient 1, extensive removal of mesh from the inguinal region resulted in a large hernia defect that developed within 3 months of the surgery. Repair of these large hernias after the extensive resection of fascia required with mesh extraction remains an unsolved problem. Rotational flaps of muscle and fascia may be indicated for relief of these large fascial defects (Figure 1).
Tumor cell entrapment in mesh and at other sites of surgical trauma
Cancer cells progressing within hernia mesh is not the only example of tumor cell entrapment [7]. Peritoneal metastases have been frequently reported in laparoscopic trocar sites following colorectal cancer resection and following a diagnostic laparoscopy in patients with malignant peritoneal mesothelioma [8,9]. It can occur in an abdominal incision after ovarian cancer surgery. Intraabdominal cancer cells can implant at a corpushemorrhagicum on the ovary and develop into a Krukenberg ovarian metastasis [10]. Any inflammatory “sticky site” within the abdomen can be a focus for cancer cell implantation and progression.
REFERENCES
10.Sugarbaker PH, Averbach AM. Krukenberg syndrome as a natural manifestation of tumor cell entrapment. In: Sugarbaker PH, editor. Peritoneal Carcinomatosis: Principles of Management. Kluwer: Boston. 1996; 163-191.